Tissue factor: newer concepts in thrombosis and its role beyond thrombosis and hemostasis
Introduction
Tissue factor (TF) is a cell surface glycoprotein normally not expressed by cells exposed to flowing blood such as endothelial cells, to prevent any improper coagulation cascade activation (1). However, it has been demonstrated that in such pathological conditions such as in acute coronary syndromes (ACS) (2) in several inflammatory diseases (3-5), in cancer (6), and in sepsis (7), TF is exposed to flowing blood. Thus, TF interaction with other coagulation factors, mainly factor VII, activates the extrinsic coagulation pathway finally leading to the acute formation of intravascular thrombus (8).
In the last two decades, our knowledge of TF biology is significantly changed (9). Specifically, in the context of coagulation, the discovery of a circulating TF pool, in which TF may be found as soluble form (10,11), bound to MPs (12,13) or blood cells-derived (14-18) has modified the old biological dogma of TF localized in the vessel-wall under the endothelial layer (19). Moreover, many studies have clearly indicated that TF, beyond the primary role as initiator of the coagulation cascade, is actively involved in other biological phenomena not related with coagulation. Indeed, it has been demonstrated that TF modulates signaling transduction, apoptosis (20), gene and protein expression (21), cell proliferation (22), angiogenesis (23,24), and tumor metastasis (23,24).This review, starting from the old point of view about TF as initiator of extrinsic coagulation pathway, briefly illustrates the more recent concepts about TF and thrombosis and finally gives an overview about its role beyond thrombosis and haemostasis focusing on the different intracellular mechanisms triggered by its activation and potentially involved in atherosclerosis.
The old dogma: TF as the key activator of the coagulation cascade
TF is a glycosylated transmembrane protein, also called coagulation factor III or tissue thromboplastin or CD142 (1). It belongs to the class 2 cytokine receptor superfamily (25). The mature protein includes 263-amino acids in a single polypeptide chain (26). The long extracellular N-terminal domain consists of 219 amino acids organized as two fibronectin type III domains, essential for factor VIIa (FVIIa) binding. The membrane anchoring peptide includes 23-amino acids while the intracellular C-terminus domain consists of 21-amino acids (27). Despite being a short peptide, the TF cytoplasmic tail has two phosphorylation sites that could be also palmitoylated (28) and/or ubiquitinated (29). In many human pathological conditions, mainly tumor metastasis, TF/FVIIa signaling involves phosphorylation of the cytoplasmatic domain at Ser253 by protein kinase C (PKC) (28,30) and at Ser258 by mitogen-activated protein kinase (MAPK) p38 (31), inducing conformational changes that may affect ligand binding (32,33).
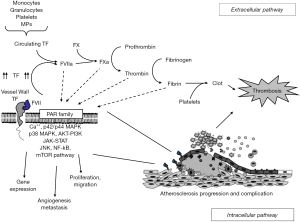
Following vessel damage, the subendothelial TF is exposed to the circulating FVII/FVIIa, which is then bound with high affinity. In this complex, the activity of FVII is enhanced of several fold due to the rearrangements of the active site region (34). To exert its full activity, the complex TF/FVIIa requires calcium and the phospholipid (PL) bilayer to activate both FX and FIX (1). In the absence of PL, the rate of activation is slow (34,35). As final step, thrombin (FIIa) is generated at the site of vessel injury thus, leading to platelet activation and aggregation, fibrin production and ultimately intravascular thrombus formation as shown in Figure 1 (1). Recent advances suggest an alternative model of coagulation initiation in which the nascent FXa formed by the complex TF-FVIIa directly activates factor VIII and factor IX, prior to inhibition by TF pathway inhibitor (TFPI) and independent of thrombin feedback. FVa generation requires FXa undocking from TF-FVIIa, thus exposing free FXa to inhibitory control by TF (36). Once constituted, the tertiary complex TF/FVIIa/Ca2+ generates FIXa and FXa. Biochemically, FXa seems to be at a critical point of the coagulation process because it receives the upstream (extrinsic and intrinsic) signals and dictates the downstream coagulation. For these reasons, in the last decade several pharmacological strategies have been developed to modulate its function in the coagulation cascade (37).
Blood-borne TF: the new dogma with a real procoagulant effect?
In 1999, Giesen et al. reported, for the first time, the presence of TF also in the circulating blood (the so-called blood-borne TF) (38). This initial observation was confirmed by many other reports; thus it is now largely accepted that TF circulates in the flowing blood at least in three different pools: as white cells and platelets-associated (16,39); as cell-derived MPs (13); and as a soluble protein (namely alternative splicing TF or asTF) (11). Most of the circulating cells, once “activated” might be a potential source of blood-borne (39). Monocytes are one of the major sources of TF (39,40), involved in thrombus formation in patients with myocardial infarction (41,42) as well as in other thrombotic disorders (15). Although TF synthesis and expression by platelets has been matter of debate (43,44), in vitro generation of platelet-like particles from differentiated human megakaryocytes showed that platelets can carry both TF protein and mRNA (45) and platelet activation can induce maturation of this TF mRNA, protein translation and surface translocation (46). TF is also expressed by neutrophils triggering thrombin generation and thrombus formation (47). Activation of neutrophils is required for TF exposure on the cell membrane (14). Platelets (16), neutrophils (48) and, as reported more recently, even T-lymphocytes (17) may be an important source of TF in patients presenting with ACS. Human eosinophils can carry TF in intracellular granules (49). Upon stimulation with platelet-activating factor (PAF) or granulocyte macrophage colony-stimulating factor, TF is translocated to cell surface, where it can trigger coagulation and supports trans endothelial migration (50). Indeed, eosinophils has been detected in thrombi from patients with in-stent thrombosis (51).
It has been postulated that blood-borne TF might be involved in thrombus growth and propagation. Specifically, this circulating TF, after binding to FVII, should amplify coagulation by “bearing” TF/FVIIa activity close to the site where platelet activation occurs. These observations deserve very important pathophysiological implications since they suggest that coagulation cascade might not be necessarily triggered by vessel injury or by atherosclerotic plaque rupture, and that other intravascular events still partially unknown might be involved (52). Fortunately, it has been demonstrated that, to avoid any improper activation of the coagulation pathway, the majority of blood-borne TF is “encrypted” and “decryption” is essential to exert its activity (53). As reported above, blood borne TF circulates in three different pools. The pool with the most important pathophysiological meaning is represented by MPs. In fact, it has been reported that elevated levels of circulating MPs are measurable in plasma of ACS patients as compared to healthy subjects or patients with stable angina (54,55). Interestingly, MPs derived from atherosclerotic plaque seem to have origin not only by cells normally involved in plaque formation such as macrophages and smooth muscle cells, but also from other blood cells such as erythrocytes, that do not have any role in atherosclerosis (54). Another type of flowing TF is the so called alternatively spliced TF (asTF) (11), that does not seem to have procoagulant activity (56).
Beyond coagulation: TF and intracellular signaling
For many years, the coagulation control has been considered the main pathophysiological role of TF. In the last two decades, several reports have clearly indicated that this glycoprotein exerts other functions besides being involved in coagulation since the TF-FVIIa complex seems able to activate many intracellular pathways as shown in Figure 1.
TF signaling: cytoplasmatic mediators
Accumulated evidence suggests that TF/FVIIa signaling may act through different intracellular mediators. The first report suggesting the possible role of TF as “true membrane receptor” has been published in 1995 showing that interaction by TF and FVIIa triggered intracellular calcium mobilization in cytokine stimulated human umbilical vein endothelial cells (HUVECs), monkey kidney fibroblasts (COS-1), human bladder carcinoma cell line J82, and canine kidney cells (MDCK) (57). Later studies have also indicated that by binding FVIIa, TF may affect gene expression in fibroblasts (58). This effect seems to be related to transient activation of p44/42 MAPK (59) and of other members of this family, such as C-Jun N-terminal kinase and p38 (60). Upstream activation of p44/42 MAPK is linked to Src family members c-Src, Lyn, and Yes, and subsequently phosphatidylinositol 3-kinase (PI3K), followed by stimulation of c-Akt/protein kinase B as well as the small GTPases Rac and Cdc42. Rac pathway mediates p38 MAPK activation and cytoskeletal reorganization (61). Interestingly, the TF/FVIIa signaling elicited by Src is dependent of the cell type. For example, in both wild type baby hamster kidney (BHK) cells and in human keratinocytes (HaCaT) cells FVIIa-induced MAP kinase activation correlates with p21Ras activation. In BHK cells, early p21Ras activation was mediated by the activation of PKC, whereas late p21Ras activation employed alternative mechanisms. In HaCaT cells, stimulation of the Src kinase family mediated FVIIa-dependent p21 Ras activation. Finally, in both cell types, Raf activity was mandatory for MAP kinase activation (62). Moreover, in BHK cells TF/FVIIa interaction also resulted in STAT5 phosphorylation and nuclear translocation and transactivation of a STAT5 reporter construct (63). It has been shown that this activation was dependent by FVIIa proteolytic activity but not by coagulation factors Xa and thrombin, or TF cytoplasmic domain. STAT5 phosphorylation mediated by TF/FVIIa was dependent on functional G12/G13 class G proteins and Jak2 activity, but not Jak1 or Tyk2. The final effect of this signaling is the Jak2/STAT5-dependent production of the antiapoptotic STAT5 target Bcl(XL) as well as Jak2-dependent activation of the antiapoptotic protein PKB (63). In fibroblasts, binding TF/FVIIa leads to activation of phospholipase C (PLC) and enhances chemotaxis (64). In human breast cancer cells, the complex TF/FVIIa promotes phosphorylation of p44/42 MAPK and Akt/protein kinase B and triggers activation of the mammalian target of rapamycin (mTOR) pathway and the phosphorylation of AKT, mTOR, andp70S6K1 (65). However, it has to be kept into account that most of these results comes from studies on immortalized cell-lines, thus caution is required in analyzing these data.
TF signaling: PARs activation
The complexes TF/FVIIa, TF/FVIIa/FXa, thrombin and activated protein C (aPC) have protease activity that cleaves the extracellular domains of the protease activated receptors (PARs) finally inducing G protein and b-arrestin coupled signaling (66). The PAR family includes four members: PAR-1, PAR-2, PAR-3 and PAR-4. Activation of PAR-1, PAR-3 and PAR-4 occurs mainly via thrombin (67), PAR-1 can be also activated by FXa (68), aPC and matrix metalloproteinase-1 (MMP-1) and MMP-13 (69). Since PAR-2 may be activated by several proteases belonging to the coagulation pathways, such as FVIIa and FXa, as well as the complex TF/FVIIa/FXa (70), it has been suggested that PAR-2 pathway plays a pivotal in modulating TF signaling, which induces phosphorylation of the cytoplasmic domain at serine residues level both in vivo and in vitro (28,30,71). Once phosphorylated, TF tail may serve as a binding site for the actin-binding protein filamin A, which is recruited to the C-terminal tail when TF/FVIIa binding occurs at extracellular level (72,73). However, TF-mediated cell signaling can thereby occurs through mechanisms related or not related to its intracellular part. For example, PAR-2 cleavage and certain proximal signaling responses of TF-FVIIa do not require the TF cytoplasmic domain (74), while it seems essential for signaling complexes and protein trafficking (i.e., the regulatory subunit of PI3K) (75), the actin binding protein filamin (72) and the prolyl-isomerase Pin1 (33). It has been recently reported that Pin1 both enhances TF gene expression via activation of NF-κB and AP-1 signaling and directly interacts with TF through a well-conserved phosphorylated Ser258-Pro259 motif in its cytoplasmic domain (33). Moreover, Pin1 increases both the protein half-life and pro-coagulant activity of TF in vascular cells. Additional effects of Pin1 on TF activity may come from PAR 2-induced release of TF on microvesicles (76).
Interaction of TF cytoplasmic domain with the regulatory subunit of PI3K is important for TF/FVIIa-PAR2 signaling connected to cell adhesion receptors, particularly integrins of the b3 and b1 families (77). It has been shown that blocking TF-integrin interaction results on one hand, in minimal effects on coagulation, but on the other hand, in inhibition of TF/FVIIa signaling and suppression of tumor growth (78). However, TF cytoplasmatic and intracellular domains seem not essential for some biological functions. These conclusions come from the studies performed on the variant of TF generated by an alternative splicing of its mRNA that creates a soluble isoform (asTF) lacking of the transmembrane and cytoplasmic domain (11). Despite of its procoagulant activity is still matter of debate (11,56), asTF retains the ability to ligate integrins (24) which might be considered as the main asTF signaling, independently of PAR2 pathway. TF integrin signaling and its implications in angiogenesis and migration has been recently further clarified. TF/FVIIa interacts with integrin through a specific binding site in the FVIIa protease domain required for PAR-2 cleavage (77). In cancer cells, TF/FVIIa-PAR2 signaling induces the Akt phosphorylation and inactivation of glycogen synthase kinase-3b (GSK-3b) that result in b-catenin stabilization and transcriptional activation of promigratory and prometastatic genes (79). In non-cancer cells, such as keratinocytes, TF/FVIIa proangiogenic signaling is integrins independent and it is associated to transactivation of epidermal growth factor receptor (EGFR) and proline-rich tyrosine kinase 2 (PYK2) (80). The TF/FVIIa complex also induces transactivation of the insulin growth factor receptor (IGF-1R) that leads to TRAIL (TNF-related apoptosis inducing ligand) inhibition, thus preventing apoptosis in cancer cells (81). Moreover, TF/FVIIa complex also induces cleavage of the tyrosine kinase receptors EphB2 and EphA2, which increases cell migration (82). Recently, TF/FVIIa complex has been linked to activation of the transmembrane serine protease matriptase that are critical enzymes in the development and homeostasis of epithelial barrier tissues (83). Transactivation of matriptase may connect coagulation cascade to epithelial defense and repair programs and contribute to pathogenic effects of extrinsic pathway activation in cancer and inflammation (83). However, further studies are warranted to clarify the role of matriptase in PAR2-independent TF/FVIIa-triggered cell surface proteolysis.
Based on the data above, it is possible to postulate: first, that activation of MAPK, PI3K-AKT, JAK-STAT and mTOR pathways links TF signaling to a multitude of functions within the cell, ranging from mitosis to migration and cell survival; second, that TF-FVIIa complex signaling through PAR2 is implicated in cancer cell migration, invasion, proliferation and evasion from apoptotic cell death.
TF cytoplasmatic domain and PAR2 signaling
The roles of TF/FVIIa-PAR2 signaling in physiology and pathology depends on the cell type and have been clarified by using experimental mice with a deletion of the TF cytoplasmic domain. In this section, we will explore the putative pathological involvement of TF/FVIIa-PAR2 signaling based on the cells type.
First, animals with TF cytoplasmatic deletion showed increased hypoxia-driven angiogenesis that was reversed by PAR2 deficiency or by inhibitors of TF-FVIIa (84,85). Based on these data and on those described in other reports (71), TF cytoplasmatic domain and PAR2-signaling seems to cooperate for a regulatory role in angiogenesis and tumor growth in breast cancer (86) as well as in hepatocellular carcinoma (87). It has been also shown that in intestine TF cytoplasmic domain participates in TF trafficking and surface localization and in cooperation with PAR1 signaling in adaptive angiogenesis following colonization of the small intestine with microbiota (88). Moreover, in adipocytes, TF cytoplasmic domain is involved in the suppression of Akt phosphorylation by insulin linked to the impairment expression of negative regulators of weight gain as supported by the studies on obese mice in which blocking TF/FVIIa interaction improves the overall metabolism (89). TF is upregulated in the obese visceral adipose tissue and expressed by adipose tissue macrophages (90). It has been reported that loss of TF/FVIIa-PAR2 signaling in hematopoietic cells prevents the development of adipose tissue inflammation and insulin resistance, thus suggesting a possible contribution to the development of type 2 diabetes and the metabolic syndrome (89). In the liver, TF/FVIIa-PAR2 signaling is linked to hepatic lipogenesis and gluconeogenesis, thus contributing to steatosis and hepatic insulin resistance (91). Animal models of hepatic inflammation show that the use of dabigatran, a recent approved thrombin inhibitor for the treatment of atrial fibrillation, reduce fibrin deposition and the related inflammation (91), supporting the notion that coagulation-induced fibrin deposition participates in inflammatory TF/FVIIa-PAR2 signaling in metabolic diseases.
The ternary complex TF/FVIIa/FXa requires endothelial protein C receptor (EPCR) for signaling in endothelial, smooth muscle and cancer cells (92). The resultant quaternary TF/FVIIa/FXa/EPCR complex participates to the innate immune response by inducing expression of the TLR3/4 signaling adaptor protein pellino-1, the transcription factor interferon (IFN) regulatory factor 8 (IRF8) and a set of IFN-regulated genes (93). This novel cooperative role of TF signaling in innate immune response is regulated by the anticoagulant PC pathway (94).
The role of TF in inflammation is, at least in part, mediated by endothelial cell, predominantly through intracellular signaling rather than coagulation activation (95). In experimental animal models, TF signaling is linked to upregulation of IL-6 without changing markers of intravascular coagulation (96). A recent report indicates that TF is regulated in endothelial cell by the anti-oxidative protein PON2, a cell-associated protein with anti-atherogenic properties (97). In experimental model of PON2 deficiency, a post-transcriptional upregulation of endothelial cell TF activity and a proinflammatory state, via expression of IL-6 and CCL22, is observed (97,98), thus linking the loss of PON2 antioxidant functions to vascular inflammation and dysfunction.
TF and signaling beyond coagulation in cardiovascular disease
The evidences that TF, besides activating coagulation cascade, has a role as a true receptor on surface of several cells have opened a new scenario in the pathophysiology of cardiovascular disease. Acute cardiovascular events occur when atherosclerosis, a chronic disease that progresses silently and often without any clinical manifestation, evolves toward acute thrombotic complications (99). The thrombogenic role of TF in these events has been well characterized over the past years (3,8,100). On the contrary, although it has been shown that TF is expressed in several human tissues (101), including healthy vascular vessel wall (102) and atherosclerotic lesions (100) it is still under investigation whether this glycoprotein might play a role in atherosclerotic plaque development and progression. It is known that proliferation and migration of smooth muscle cells is an important step in atherosclerotic plaque grow and stability (103). Previous report has indicated that binding TF by human recombinant FVIIa induces cell proliferation via activation of p44/42 and its translocation to the nucleus (22). Moreover, binding of FVII to its natural receptor, TF, activates the Wnt/β-catenin pathway, with Rho GTPases as key mediators, finally leading to migration of other cells normally represented in the vessel wall such as endothelial cells (85). It is known that plaque neovascularization may have a crucial role in plaque instability (104), and this phenomenon angiogenesis is a crucial mechanism for oxygen supply of the growing plaque contents (104). Several chemical mediators such as VEGF, HIF-1α and EGR-1 are involved in this step (104). The role of TF as trigger for several intracellular pathways involving these chemical mediators has been well documented (85).
Atherosclerosis is considered an inflammatory disease (105-107). In this context, the TF/FVII complex, thru PAR-2 pathway activation, activates several proinflammatory signaling (108,109), that, in turn, cause secretion of inflammatory cytokines, such as IL-6 and other chemokines, finally amplifying the atherogenic process (110). Thus TF seems to have a crucial role in initiation and amplification of the atherosclerotic process too (111).
Taken together, these data suggest that development, as well as progression of atherosclerotic disease might be, in part, due to the TF activity as a true receptor able to trigger several molecular pathways that, in turn, lead to plaque grow and instability by mediating cellular proliferation and migration, angiogenesis and inflammation.
TF: potential therapeutic approaches?
The demonstrations that the TF/FVIIa complex may exert other functions, besides being involved in coagulation, have opened several hypothetical interesting therapeutic scenarios. However, it is extremely complicated to modulate separately the two TF activity (active effector/true receptor). It should be kept in mind that TF main physiological activity is to protect against bleeding, while its pathological role could be considered evident only in several diseases, such as atherosclerosis and, mainly, ACSs. Thus, the fine balance between these two TF activities (protective and pathological functions) appears to be the great limitation in terms of therapeutic strategies.
It has been suggested that a potential therapeutic approach to balance risks and benefits might be represented by selective modulation of TF/FVIIa pathway (112,113), including PAR signaling (114) and/or the proteases generated during the coagulation cascade (37,115). To date, preclinical studies with different anti-TF interventions have been conducted with promising results in terms of antithrombotic efficacy (116), anti-angiogenetic effects (117), as well as anti-inflammatory (118) and anti-fibrotic potential (119). However, despite these promising results, the clinical trial was designed to contrast only the procoagulant TF effects and failed to confirm the safety of TF inhibition, because an increased risk of major bleeding and, consequently, of mortality was observed in the treated group (120). More promising results come from the only physiological inhibitor of TF/FVIIa complex, TFPI (121). It is synthesized and released by endothelial cells and binds to the complex TF/FVIIa/FXa to inhibit the downstream pathway that ends with thrombin activation and clot formation (122). Several studies have underlined that recombinant human TFPI could have anti-inflammatory effects and inhibits cell migration as well as progression of atherosclerosis (123). Active site-inhibited FVIIa (FFR-FVIIa) is another possible therapeutic inhibitor of the TF-FVIIa-dependent initiation of coagulation. It competes with endogenous FVIIa for binding to TF and, thus limiting the formation of functional TF-FVIIa complex (124). Its possible use as therapeutic agent to reduce thrombus formation and inflammatory changes has been already reported (125). Other small molecule inhibitors, namely naphthylamidines, have been tested in vivo and shown to be efficacious in inhibiting thrombus formation and reducing bleeding (126). However, because of a non-specific inhibition of other coagulation proteases aside from inhibition of FVIIa activity, their use has been limited. Anti-TF antibodies have been also tested as anti-thrombotic agents, designed to interfere with the association of TF with FVIIa (127) or with substrate docking (128). Exogenous inhibitors of the TF/FVIIa catalytic complex have been isolated from hookworms. Nematode anticoagulant protein c2 (rNAPc2) is an 85-amino-acid serine protease inhibitor that in contrast to TFPI binds FX or FXa prior to the formation of an inhibitory complex with TF/FVIIa (129). Its anticoagulant effect has been investigated in ischemic patients (130). Recently, novel and more sophisticated approaches have been identified based on the complex biology of TF. Targeting specific post-transcriptional modifications of the TF gene, such as alternative splicing, may be potentially addressable (131). The recent discovery of the role of non-coding microRNA in modulating biological functions (132,133) opened a new and interesting scenario. For example, it has been already reported that microRNA-19b, that functions downregulating TF mRNA, may exert potential anti-thrombotic properties in patients with unstable angina (134).
It must be pointed out that most of these agents has been designed to test the anticoagulant properties and to prevent thrombosis. However, beyond coagulation, TF is involved in many intracellular signaling that linked to different pathological disorders. It has been reported that in addition to inhibition of coagulation anti-TF antibodies and NAPc2 may reduce tumour growth and metastasis by interfering with signalling pathways (135,136).
Recently, a first generation of TF-Targeting Antibody-Like Immunoconjugates (called ICON or ICON-1) has been investigated in cancer disease. ICON is a 210 kDa chimeric antibody-like homodimer that consists of murine or human FVII fused to the Fc region of IgG1. The procoagulant effects of ICON-encoded zymogen FVII have been significantly eliminated via targeted mutation of the lysine reside at position 341 (K341A) (137). Its administration has been associated to marked tumor inhibition, and in some cases, complete eradication without affecting normal tissues. Upon binding to TF-expressing malignant cells, ICON can mediate natural killer and complement-dependent cytotoxicity (CDC) as its mechanism of action (137). However, because the procoagulant activity in ICON was not completely eliminated (138) coagulation disorder may occur in cancer patients who usually have a hypercoagulation status. Starting from this limitation, a second-generation ICON, named L-ICON1, consisting of only the light chain of FVII fused to an IgG1 Fc has been developed (139). These newer and improved TF-targeting agents are being evaluated in preclinical studies with potential to translate into clinical trials.
Since translation of preclinical studies in clinical practice is always difficult, large randomized trials addressing hard endpoints should be designed to confirm the potential therapeutic role of TF/FVIIa inhibition. In this context, however, it should be remarked that any new strategy has a therapeutic role its benefit will overcome any side effect.
Conclusions
In conclusion, TF should be no more considered as the trigger of extrinsic coagulation pathway only. Recent evidences demonstrating that it is a true receptor involved in several cellular mechanisms, have indicated that this glycoprotein might be involved in a wider number of pathophysiological phenomena beyond coagulation. Starting from these new concepts, researcher have focused their attention to this glycoprotein by another point of view and have done many efforts to obtain a clearer point of view about the role played by this molecule in several disease. Thus, several potential therapeutic approaches have been proposed including those to reduce, at the same time, the atherosclerosis progression and its acute complication in a thrombotic acute event.
Acknowledgements
This report is dedicated to my family, Lucia, Carmen and Marco for their continuous support.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Cimmino G, Ciccarelli G, Golino P. Role of Tissue Factor in the Coagulation Network. Semin Thromb Hemost 2015;41:708-17. [Crossref] [PubMed]
- Breitenstein A, Tanner FC, Luscher TF. Tissue factor and cardiovascular disease: quo vadis? Circ J 2010;74:3-12. [Crossref] [PubMed]
- Cimmino G, D’Amico C, Vaccaro V, et al. The missing link between atherosclerosis, inflammation and thrombosis: is it tissue factor? Expert Rev Cardiovasc Ther 2011;9:517-23. [Crossref] [PubMed]
- Meerarani P, Moreno PR, Cimmino G, et al. Atherothrombosis: role of tissue factor; link between diabetes, obesity and inflammation. Indian J Exp Biol 2007;45:103-10. [PubMed]
- Cirillo P, Golino P, Calabro P, et al. C-reactive protein induces tissue factor expression and promotes smooth muscle and endothelial cell proliferation. Cardiovasc Res 2005;68:47-55. [Crossref] [PubMed]
- Ruf W. Tissue factor and cancer. Thromb Res 2012;130 Suppl 1:S84-7. [Crossref] [PubMed]
- Pawlinski R, Mackman N. Cellular sources of tissue factor in endotoxemia and sepsis. Thromb Res 2010;125 Suppl 1:S70-3. [Crossref] [PubMed]
- Toschi V, Gallo R, Lettino M, et al. Tissue factor modulates the thrombogenicity of human atherosclerotic plaques. Circulation 1997;95:594-9. [Crossref] [PubMed]
- Cimmino G, Fischetti S, Golino P. The Two Faces of Thrombosis: Coagulation Cascade and Platelet Aggregation. Are Platelets the Main Therapeutic Target? J Thrombo Cir 2017;3:117.
- Szotowski B, Antoniak S, Poller W, et al. Procoagulant soluble tissue factor is released from endothelial cells in response to inflammatory cytokines. Circ Res 2005;96:1233-9. [Crossref] [PubMed]
- Bogdanov VY, Balasubramanian V, Hathcock J, et al. Alternatively spliced human tissue factor: a circulating, soluble, thrombogenic protein. Nat Med 2003;9:458-62. [Crossref] [PubMed]
- Davizon P, Lopez JA. Microparticles and thrombotic disease. Curr Opin Hematol 2009;16:334-41. [Crossref] [PubMed]
- Lechner D, Weltermann A. Circulating tissue factor-exposing microparticles. Thromb Res 2008;122 Suppl 1:S47-54. [Crossref] [PubMed]
- Maugeri N, Manfredi AA. Tissue Factor Expressed by Neutrophils: Another Piece in the Vascular Inflammation Puzzle. Semin Thromb Hemost 2015;41:728-36. [Crossref] [PubMed]
- Shantsila E, Lip GY. The role of monocytes in thrombotic disorders. Insights from tissue factor, monocyte-platelet aggregates and novel mechanisms. Thromb Haemost 2009;102:916-24. [Crossref] [PubMed]
- Brambilla M, Camera M, Colnago D, et al. Tissue factor in patients with acute coronary syndromes: expression in platelets, leukocytes, and platelet-leukocyte aggregates. Arterioscler Thromb Vasc Biol 2008;28:947-53. [Crossref] [PubMed]
- De Palma R, Cirillo P, Ciccarelli G, et al. Expression of functional tissue factor in activated T-lymphocytes in vitro and in vivo: A possible contribution of immunity to thrombosis? Int J Cardiol 2016;218:188-95. [Crossref] [PubMed]
- Camera M, Toschi V, Brambilla M, et al. The Role of Tissue Factor in Atherothrombosis and Coronary Artery Disease: Insights into Platelet Tissue Factor. Semin Thromb Hemost 2015;41:737-46. [Crossref] [PubMed]
- Cimmino G, Golino P, Badimon JJ. Pathophysiological role of blood-borne tissue factor: should the old paradigm be revisited? Intern Emerg Med 2011;6:29-34. [Crossref] [PubMed]
- Aberg M, Siegbahn A. Tissue factor non-coagulant signaling - molecular mechanisms and biological consequences with a focus on cell migration and apoptosis. J Thromb Haemost 2013;11:817-25. [Crossref] [PubMed]
- Chu AJ. Tissue factor, blood coagulation, and beyond: an overview. Int J Inflam 2011;2011. [Crossref] [PubMed]
- Cirillo P, Cali G, Golino P, et al. Tissue factor binding of activated factor VII triggers smooth muscle cell proliferation via extracellular signal-regulated kinase activation. Circulation 2004;109:2911-6. [Crossref] [PubMed]
- van den Berg YW, Versteeg HH. Alternatively spliced tissue factor. A crippled protein in coagulation or a key player in non-haemostatic processes? Hamostaseologie 2010;30:144-9. [Crossref] [PubMed]
- van den Berg YW, van den Hengel LG, Myers HR, et al. Alternatively spliced tissue factor induces angiogenesis through integrin ligation. Proc Natl Acad Sci U S A 2009;106:19497-502. [Crossref] [PubMed]
- Bazan JF. Structural design and molecular evolution of a cytokine receptor superfamily. Proc Natl Acad Sci U S A 1990;87:6934-8. [Crossref] [PubMed]
- Broze GJ Jr, Leykam JE, Schwartz BD, et al. Purification of human brain tissue factor. J Biol Chem 1985;260:10917-20. [PubMed]
- Sharma L, Melis E, Hickey MJ, et al. The cytoplasmic domain of tissue factor contributes to leukocyte recruitment and death in endotoxemia. Am J Pathol 2004;165:331-40. [Crossref] [PubMed]
- Dorfleutner A, Ruf W. Regulation of tissue factor cytoplasmic domain phosphorylation by palmitoylation. Blood 2003;102:3998-4005. [Crossref] [PubMed]
- Ettelaie C, Collier ME, Featherby S, et al. Oligoubiquitination of tissue factor on Lys255 promotes Ser253-dephosphorylation and terminates TF release. Biochim Biophys Acta 2016;1863:2846-57. [Crossref] [PubMed]
- Ahamed J, Ruf W. Protease-activated receptor 2-dependent phosphorylation of the tissue factor cytoplasmic domain. J Biol Chem 2004;279:23038-44. [Crossref] [PubMed]
- Ettelaie C, Elkeeb AM, Maraveyas A, et al. p38alpha phosphorylates serine 258 within the cytoplasmic domain of tissue factor and prevents its incorporation into cell-derived microparticles. Biochim Biophys Acta 2013;1833:613-21. [Crossref] [PubMed]
- Sen M, Herzik M, Craft JW, et al. Spectroscopic Characterization of Successive Phosphorylation of the Tissue Factor Cytoplasmic Region. Open Spectrosc J 2009;3:58-64. [Crossref] [PubMed]
- Kurakula K, Koenis DS, Herzik MA Jr, et al. Structural and cellular mechanisms of peptidyl-prolyl isomerase Pin1-mediated enhancement of Tissue Factor gene expression, protein half-life, and pro-coagulant activity. Haematologica 2018;103:1073-82. [Crossref] [PubMed]
- Sabharwal AK, Birktoft JJ, Gorka J, et al. High affinity Ca(2+)-binding site in the serine protease domain of human factor VIIa and its role in tissue factor binding and development of catalytic activity. J Biol Chem 1995;270:15523-30. [Crossref] [PubMed]
- Ruf W, Rehemtulla A, Morrissey JH, et al. Phospholipid-independent and -dependent interactions required for tissue factor receptor and cofactor function. J Biol Chem 1991;266:2158-66. [PubMed]
- Schuijt TJ, Bakhtiari K, Daffre S, et al. Factor Xa activation of factor V is of paramount importance in initiating the coagulation system: lessons from a tick salivary protein. Circulation 2013;128:254-66. [Crossref] [PubMed]
- Borensztajn K, Spek CA. Blood coagulation factor Xa as an emerging drug target. Expert Opin Ther Targets 2011;15:341-9. [Crossref] [PubMed]
- Giesen PL, Rauch U, Bohrmann B, et al. Blood-borne tissue factor: another view of thrombosis. Proc Natl Acad Sci U S A 1999;96:2311-5. [Crossref] [PubMed]
- Osterud B, Bjorklid E. Sources of tissue factor. Semin Thromb Hemost 2006;32:11-23. [Crossref] [PubMed]
- Peshkova AD, Le Minh G, Tutwiler V, et al. Activated Monocytes Enhance Platelet-Driven Contraction of Blood Clots via Tissue Factor Expression. Sci Rep 2017;7:5149. [Crossref] [PubMed]
- Chiva-Blanch G, Laake K, Myhre P, et al. Platelet-, monocyte-derived and tissue factor-carrying circulating microparticles are related to acute myocardial infarction severity. PLoS One 2017;12. [Crossref] [PubMed]
- Leatham EW, Bath PM, Tooze JA, et al. Increased monocyte tissue factor expression in coronary disease. Br Heart J 1995;73:10-3. [Crossref] [PubMed]
- Camera M, Brambilla M, Toschi V, et al. Tissue factor expression on platelets is a dynamic event. Blood 2010;116:5076-7. [Crossref] [PubMed]
- Bouchard BA, Mann KG, Butenas S. No evidence for tissue factor on platelets. Blood 2010;116:854-5. [Crossref] [PubMed]
- Brambilla M, Facchinetti L, Canzano P, et al. Human megakaryocytes confer tissue factor to a subset of shed platelets to stimulate thrombin generation. Thromb Haemost 2015;114:579-92. [Crossref] [PubMed]
- Schwertz H, Tolley ND, Foulks JM, et al. Signal-dependent splicing of tissue factor pre-mRNA modulates the thrombogenicity of human platelets. J Exp Med 2006;203:2433-40. [Crossref] [PubMed]
- Darbousset R, Thomas GM, Mezouar S, et al. Tissue factor-positive neutrophils bind to injured endothelial wall and initiate thrombus formation. Blood 2012;120:2133-43. [Crossref] [PubMed]
- Stakos DA, Kambas K, Konstantinidis T, et al. Expression of functional tissue factor by neutrophil extracellular traps in culprit artery of acute myocardial infarction. Eur Heart J 2015;36:1405-14. [Crossref] [PubMed]
- Cugno M, Marzano AV, Lorini M, et al. Enhanced tissue factor expression by blood eosinophils from patients with hypereosinophilia: a possible link with thrombosis. PLoS One 2014;9. [Crossref] [PubMed]
- Moosbauer C, Morgenstern E, Cuvelier SL, et al. Eosinophils are a major intravascular location for tissue factor storage and exposure. Blood 2007;109:995-1002. [Crossref] [PubMed]
- Riegger J, Byrne RA, Joner M, et al. Histopathological evaluation of thrombus in patients presenting with stent thrombosis. A multicenter European study: a report of the prevention of late stent thrombosis by an interdisciplinary global European effort consortium. Eur Heart J 2016;37:1538-49. [Crossref] [PubMed]
- Balasubramanian V, Grabowski E, Bini A, et al. Platelets, circulating tissue factor, and fibrin colocalize in ex vivo thrombi: real-time fluorescence images of thrombus formation and propagation under defined flow conditions. Blood 2002;100:2787-92. [Crossref] [PubMed]
- Bach RR. Tissue factor encryption. Arterioscler Thromb Vasc Biol 2006;26:456-61. [Crossref] [PubMed]
- Leroyer AS, Isobe H, Leseche G, et al. Cellular origins and thrombogenic activity of microparticles isolated from human atherosclerotic plaques. J Am Coll Cardiol 2007;49:772-7. [Crossref] [PubMed]
- Mallat Z, Hugel B, Ohan J, et al. Shed membrane microparticles with procoagulant potential in human atherosclerotic plaques: a role for apoptosis in plaque thrombogenicity. Circulation 1999;99:348-53. [Crossref] [PubMed]
- Censarek P, Bobbe A, Grandoch M, et al. Alternatively spliced human tissue factor (asHTF) is not pro-coagulant. Thromb Haemost 2007;97:11-4. [Crossref] [PubMed]
- Rottingen JA, Enden T, Camerer E, et al. Binding of human factor VIIa to tissue factor induces cytosolic Ca2+ signals in J82 cells, transfected COS-1 cells, Madin-Darby canine kidney cells and in human endothelial cells induced to synthesize tissue factor. J Biol Chem 1995;270:4650-60. [Crossref] [PubMed]
- Pendurthi UR, Alok D, Rao LV. Binding of factor VIIa to tissue factor induces alterations in gene expression in human fibroblast cells: up-regulation of poly(A) polymerase. Proc Natl Acad Sci U S A 1997;94:12598-603. [Crossref] [PubMed]
- Poulsen LK, Jacobsen N, Sorensen BB, et al. Signal transduction via the mitogen-activated protein kinase pathway induced by binding of coagulation factor VIIa to tissue factor. J Biol Chem 1998;273:6228-32. [Crossref] [PubMed]
- Ott I, Weigand B, Michl R, et al. Tissue factor cytoplasmic domain stimulates migration by activation of the GTPase Rac1 and the mitogen-activated protein kinase p38. Circulation 2005;111:349-55. [Crossref] [PubMed]
- Versteeg HH, Hoedemaeker I, Diks SH, et al. Factor VIIa/tissue factor-induced signaling via activation of Src-like kinases, phosphatidylinositol 3-kinase, and Rac. J Biol Chem 2000;275:28750-6. [Crossref] [PubMed]
- Versteeg HH, Bresser HL, Spek CA, et al. Regulation of the p21Ras-MAP kinase pathway by factor VIIa. J Thromb Haemost 2003;1:1012-8. [Crossref] [PubMed]
- Versteeg HH, Spek CA, Slofstra SH, et al. FVIIa:TF induces cell survival via G12/G13-dependent Jak/STAT activation and BclXL production. Circ Res 2004;94:1032-40. [Crossref] [PubMed]
- Siegbahn A, Johnell M, Rorsman C, et al. Binding of factor VIIa to tissue factor on human fibroblasts leads to activation of phospholipase C and enhanced PDGF-BB-stimulated chemotaxis. Blood 2000;96:3452-8. [PubMed]
- Jiang X, Zhu S, Panetti TS, et al. Formation of tissue factor-factor VIIa-factor Xa complex induces activation of the mTOR pathway which regulates migration of human breast cancer cells. Thromb Haemost 2008;100:127-33. [Crossref] [PubMed]
- Hamilton JR, Trejo J. Challenges and Opportunities in Protease-Activated Receptor Drug Development. Annu Rev Pharmacol Toxicol 2017;57:349-73. [Crossref] [PubMed]
- Coughlin SR. Thrombin signalling and protease-activated receptors. Nature 2000;407:258-64. [Crossref] [PubMed]
- Camerer E, Kataoka H, Kahn M, et al. Genetic evidence that protease-activated receptors mediate factor Xa signaling in endothelial cells. J Biol Chem 2002;277:16081-7. [Crossref] [PubMed]
- Austin KM, Covic L, Kuliopulos A. Matrix metalloproteases and PAR1 activation. Blood 2013;121:431-9. [Crossref] [PubMed]
- Camerer E, Huang W, Coughlin SR. Tissue factor- and factor X-dependent activation of protease-activated receptor 2 by factor VIIa. Proc Natl Acad Sci U S A 2000;97:5255-60. [Crossref] [PubMed]
- Ryden L, Grabau D, Schaffner F, et al. Evidence for tissue factor phosphorylation and its correlation with protease-activated receptor expression and the prognosis of primary breast cancer. Int J Cancer 2010;126:2330-40. [PubMed]
- Ott I, Fischer EG, Miyagi Y, et al. A role for tissue factor in cell adhesion and migration mediated by interaction with actin-binding protein 280. J Cell Biol 1998;140:1241-53. [Crossref] [PubMed]
- Koizume S, Ito S, Yoshioka Y, et al. High-level secretion of tissue factor-rich extracellular vesicles from ovarian cancer cells mediated by filamin-A and protease-activated receptors. Thromb Haemost 2016;115:299-310. [Crossref] [PubMed]
- Camerer E, Rottingen JA, Gjernes E, et al. Coagulation factors VIIa and Xa induce cell signaling leading to up-regulation of the egr-1 gene. J Biol Chem 1999;274:32225-33. [Crossref] [PubMed]
- Wang X, Gjernes E, Prydz H. Factor VIIa induces tissue factor-dependent up-regulation of interleukin-8 in a human keratinocyte line. J Biol Chem 2002;277:23620-6. [Crossref] [PubMed]
- Ettelaie C, Collier MEW, Featherby S, et al. Peptidyl-prolyl isomerase 1 (Pin1) preserves the phosphorylation state of tissue factor and prolongs its release within microvesicles. Biochim Biophys Acta Mol Cell Res 2018;1865:12-24. [Crossref] [PubMed]
- Rothmeier AS, Liu E, Chakrabarty S, et al. Identification of the integrin-binding site on coagulation factor VIIa required for proangiogenic PAR2 signaling. Blood 2018;131:674-85. [Crossref] [PubMed]
- Versteeg HH, Schaffner F, Kerver M, et al. Inhibition of tissue factor signaling suppresses tumor growth. Blood 2008;111:190-9. [Crossref] [PubMed]
- Roy A, Ansari SA, Das K, et al. Coagulation factor VIIa-mediated protease-activated receptor 2 activation leads to beta-catenin accumulation via the AKT/GSK3beta pathway and contributes to breast cancer progression. J Biol Chem 2017;292:13688-701. [Crossref] [PubMed]
- Wiiger MT, Prydz H. The epidermal growth factor receptor (EGFR) and proline rich tyrosine kinase 2 (PYK2) are involved in tissue factor dependent factor VIIa signalling in HaCaT cells. Thromb Haemost 2004;92:13-22. [Crossref] [PubMed]
- Aberg M, Eriksson O, Mokhtari D, et al. Tissue factor/factor VIIa induces cell survival and gene transcription by transactivation of the insulin-like growth factor 1 receptor. Thromb Haemost 2014;111:748-60. [Crossref] [PubMed]
- Eriksson O, Ramstrom M, Hornaeus K, et al. The Eph tyrosine kinase receptors EphB2 and EphA2 are novel proteolytic substrates of tissue factor/coagulation factor VIIa. J Biol Chem 2014;289:32379-91. [Crossref] [PubMed]
- Le Gall SM, Szabo R, Lee M, et al. Matriptase activation connects tissue factor-dependent coagulation initiation to epithelial proteolysis and signaling. Blood 2016;127:3260-9. [Crossref] [PubMed]
- Uusitalo-Jarvinen H, Kurokawa T, Mueller BM, et al. Role of protease activated receptor 1 and 2 signaling in hypoxia-induced angiogenesis. Arterioscler Thromb Vasc Biol 2007;27:1456-62. [Crossref] [PubMed]
- Belting M, Dorrell MI, Sandgren S, et al. Regulation of angiogenesis by tissue factor cytoplasmic domain signaling. Nat Med 2004;10:502-9. [Crossref] [PubMed]
- Schaffner F, Versteeg HH, Schillert A, et al. Cooperation of tissue factor cytoplasmic domain and PAR2 signaling in breast cancer development. Blood 2010;116:6106-13. [Crossref] [PubMed]
- Chen KD, Wang CC, Tsai MC, et al. Interconnections between autophagy and the coagulation cascade in hepatocellular carcinoma. Cell Death Dis 2014;5. [Crossref] [PubMed]
- Reinhardt C, Bergentall M, Greiner TU, et al. Tissue factor and PAR1 promote microbiota-induced intestinal vascular remodelling. Nature 2012;483:627-31. [Crossref] [PubMed]
- Badeanlou L, Furlan-Freguia C, Yang G, et al. Tissue factor-protease-activated receptor 2 signaling promotes diet-induced obesity and adipose inflammation. Nat Med 2011;17:1490-7. [Crossref] [PubMed]
- Samad F, Pandey M, Loskutoff DJ. Tissue factor gene expression in the adipose tissues of obese mice. Proc Natl Acad Sci U S A 1998;95:7591-6. [Crossref] [PubMed]
- Wang J, Chakrabarty S, Bui Q, et al. Hematopoietic tissue factor-protease-activated receptor 2 signaling promotes hepatic inflammation and contributes to pathways of gluconeogenesis and steatosis in obese mice. Am J Pathol 2015;185:524-35. [Crossref] [PubMed]
- Disse J, Petersen HH, Larsen KS, et al. The endothelial protein C receptor supports tissue factor ternary coagulation initiation complex signaling through protease-activated receptors. J Biol Chem 2011;286:5756-67. [Crossref] [PubMed]
- Rallabhandi P, Nhu QM, Toshchakov VY, et al. Analysis of proteinase-activated receptor 2 and TLR4 signal transduction: a novel paradigm for receptor cooperativity. J Biol Chem 2008;283:24314-25. [Crossref] [PubMed]
- Liang HP, Kerschen EJ, Basu S, et al. Coagulation factor V mediates inhibition of tissue factor signaling by activated protein C in mice. Blood 2015;126:2415-23. [Crossref] [PubMed]
- Egorina EM, Sovershaev MA, Hansen JB. The role of tissue factor in systemic inflammatory response syndrome. Blood Coagul Fibrinolysis 2011;22:451-6. [Crossref] [PubMed]
- Chantrathammachart P, Mackman N, Sparkenbaugh E, et al. Tissue factor promotes activation of coagulation and inflammation in a mouse model of sickle cell disease. Blood 2012;120:636-46. [Crossref] [PubMed]
- Ebert J, Wilgenbus P, Teiber JF, et al. Paraoxonase-2 regulates coagulation activation through endothelial tissue factor. Blood 2018;131:2161-72. [Crossref] [PubMed]
- Liang HP, Kerschen EJ, Hernandez I, et al. EPCR-dependent PAR2 activation by the blood coagulation initiation complex regulates LPS-triggered interferon responses in mice. Blood 2015;125:2845-54. [Crossref] [PubMed]
- Cimmino G, Loffredo FS, Morello A, et al. Immune-inflammatory Activation in Acute Coronary Syndromes: A Look into the Heart of Unstable Coronary Plaque. Curr Cardiol Rev 2017;13:110-7. [Crossref] [PubMed]
- Ardissino D, Merlini PA, Bauer KA, et al. Thrombogenic potential of human coronary atherosclerotic plaques. Blood 2001;98:2726-9. [Crossref] [PubMed]
- Drake TA, Morrissey JH, Edgington TS. Selective cellular expression of tissue factor in human tissues. Implications for disorders of hemostasis and thrombosis. Am J Pathol 1989;134:1087-97. [PubMed]
- Wilcox JN, Smith KM, Schwartz SM, et al. Localization of tissue factor in the normal vessel wall and in the atherosclerotic plaque. Proc Natl Acad Sci U S A 1989;86:2839-43. [Crossref] [PubMed]
- Badimon JJ, Ibanez B, Cimmino G. Genesis and dynamics of atherosclerotic lesions: implications for early detection. Cerebrovasc Dis 2009;27 Suppl 1:38-47. [Crossref] [PubMed]
- Camare C, Pucelle M, Negre-Salvayre A, et al. Angiogenesis in the atherosclerotic plaque. Redox Biol 2017;12:18-34. [Crossref] [PubMed]
- Schaftenaar F, Frodermann V, Kuiper J, et al. Atherosclerosis: the interplay between lipids and immune cells. Curr Opin Lipidol 2016;27:209-15. [Crossref] [PubMed]
- Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature 2011;473:317-25. [Crossref] [PubMed]
- Weber C, Noels H. Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 2011;17:1410-22. [Crossref] [PubMed]
- Antoniak S, Pawlinski R, Mackman N. Protease-activated receptors and myocardial infarction. IUBMB Life 2011;63:383-9. [Crossref] [PubMed]
- Borissoff JI, Spronk HM, ten Cate H. The hemostatic system as a modulator of atherosclerosis. N Engl J Med 2011;364:1746-60. [Crossref] [PubMed]
- Demetz G, Seitz I, Stein A, et al. Tissue Factor-Factor VIIa complex induces cytokine expression in coronary artery smooth muscle cells. Atherosclerosis 2010;212:466-71. [Crossref] [PubMed]
- D'Alessandro E, Posma JJN, Spronk HMH, et al. Tissue factor (Factor VIIa) in the heart and vasculature: More than an envelope. Thromb Res 2018;168:130-7. [Crossref] [PubMed]
- Golino P, Ragni M, Cirillo P, et al. Antithrombotic effects of recombinant human, active site-blocked factor VIIa in a rabbit model of recurrent arterial thrombosis. Circ Res 1998;82:39-46. [Crossref] [PubMed]
- Arnljots B, Ezban M, Hedner U. Prevention of experimental arterial thrombosis by topical administration of active site-inactivated factor VIIa. J Vasc Surg 1997;25:341-6. [Crossref] [PubMed]
- Yau MK, Liu L, Fairlie DP. Toward drugs for protease-activated receptor 2 (PAR2). J Med Chem 2013;56:7477-97. [Crossref] [PubMed]
- Alexander JH, Singh KP. Inhibition of Factor Xa: a potential target for the development of new anticoagulants. Am J Cardiovasc Drugs 2005;5:279-90. [Crossref] [PubMed]
- Priestley ES. Tissue factor-fVIIa inhibition: update on an unfinished quest for a novel oral antithrombotic. Drug Discov Today 2014;19:1440-4. [Crossref] [PubMed]
- Hu Z, Cheng J, Xu J, et al. Tissue factor is an angiogenic-specific receptor for factor VII-targeted immunotherapy and photodynamic therapy. Angiogenesis 2017;20:85-96. [Crossref] [PubMed]
- Arnold CS, Parker C, Upshaw R, et al. The antithrombotic and anti-inflammatory effects of BCX-3607, a small molecule tissue factor/factor VIIa inhibitor. Thromb Res 2006;117:343-9. [Crossref] [PubMed]
- Dhar A, Sadiq F, Anstee QM, et al. Thrombin and factor Xa link the coagulation system with liver fibrosis. BMC Gastroenterol 2018;18:60. [Crossref] [PubMed]
- Vincent JL, Artigas A, Petersen LC, et al. A multicenter, randomized, double-blind, placebo-controlled, dose-escalation trial assessing safety and efficacy of active site inactivated recombinant factor VIIa in subjects with acute lung injury or acute respiratory distress syndrome. Crit Care Med 2009;37:1874-80. [Crossref] [PubMed]
- Golino P, Ragni M, Cimmino G, et al. Role of tissue factor pathway inhibitor in the regulation of tissue factor-dependent blood coagulation. Cardiovasc Drug Rev 2002;20:67-80. [Crossref] [PubMed]
- Lwaleed BA, Bass PS. Tissue factor pathway inhibitor: structure, biology and involvement in disease. J Pathol 2006;208:327-39. [Crossref] [PubMed]
- Van Den Boogaard FE, Brands X, Schultz MJ, et al. Recombinant human tissue factor pathway inhibitor exerts anticoagulant, anti-inflammatory and antimicrobial effects in murine pneumococcal pneumonia. J Thromb Haemost 2011;9:122-32. [Crossref] [PubMed]
- Golino P, Ragni M, Cirillo P, et al. Effects of recombinant active site-blocked activated factor VII in rabbit models of carotid stenosis and myocardial infarction. Blood Coagul Fibrinolysis 2000;11 Suppl 1:S149-58. [Crossref] [PubMed]
- Lev EI, Marmur JD, Zdravkovic M, et al. Antithrombotic effect of tissue factor inhibition by inactivated factor VIIa: an ex vivo human study. Arterioscler Thromb Vasc Biol 2002;22:1036-41. [Crossref] [PubMed]
- Suleymanov OD, Szalony JA, Salyers AK, et al. Pharmacological interruption of acute thrombus formation with minimal hemorrhagic complications by a small molecule tissue factor/factor VIIa inhibitor: comparison to factor Xa and thrombin inhibition in a nonhuman primate thrombosis model. J Pharmacol Exp Ther 2003;306:1115-21. [Crossref] [PubMed]
- Himber J, Kirchhofer D, Riederer M, et al. Dissociation of antithrombotic effect and bleeding time prolongation in rabbits by inhibiting tissue factor function. Thromb Haemost 1997;78:1142-9. [Crossref] [PubMed]
- Kirchhofer D, Moran P, Chiang N, et al. Epitope location on tissue factor determines the anticoagulant potency of monoclonal anti-tissue factor antibodies. Thromb Haemost 2000;84:1072-81. [Crossref] [PubMed]
- Lee AY, Vlasuk GP. Recombinant nematode anticoagulant protein c2 and other inhibitors targeting blood coagulation factor VIIa/tissue factor. J Intern Med 2003;254:313-21. [Crossref] [PubMed]
- Moons AH, Peters RJ, Bijsterveld NR, et al. Recombinant nematode anticoagulant protein c2, an inhibitor of the tissue factor/factor VIIa complex, in patients undergoing elective coronary angioplasty. J Am Coll Cardiol 2003;41:2147-53. [Crossref] [PubMed]
- Eisenreich A, Malz R, Pepke W, et al. Role of the phosphatidylinositol 3-kinase/protein kinase B pathway in regulating alternative splicing of tissue factor mRNA in human endothelial cells. Circ J 2009;73:1746-52. [Crossref] [PubMed]
- Cimmino G, Tarallo R, Nassa G, et al. Activating stimuli induce platelet microRNA modulation and proteome reorganisation. Thromb Haemost 2015;114:96-108. [Crossref] [PubMed]
- Bhaskaran M, Mohan M. MicroRNAs: history, biogenesis, and their evolving role in animal development and disease. Vet Pathol 2014;51:759-74. [Crossref] [PubMed]
- Li S, Ren J, Xu N, et al. MicroRNA-19b functions as potential anti-thrombotic protector in patients with unstable angina by targeting tissue factor. J Mol Cell Cardiol 2014;75:49-57. [Crossref] [PubMed]
- Hembrough TA, Swartz GM, Papathanassiu A, et al. Tissue factor/factor VIIa inhibitors block angiogenesis and tumor growth through a nonhemostatic mechanism. Cancer Res 2003;63:2997-3000. [PubMed]
- Mueller BM, Reisfeld RA, Edgington TS, et al. Expression of tissue factor by melanoma cells promotes efficient hematogenous metastasis. Proc Natl Acad Sci U S A 1992;89:11832-6. [Crossref] [PubMed]
- Hu Z, Li J. Natural killer cells are crucial for the efficacy of Icon (factor VII/human IgG1 Fc) immunotherapy in human tongue cancer. BMC Immunol 2010;11:49. [Crossref] [PubMed]
- Hu Z, Garen A. Targeting tissue factor on tumor vascular endothelial cells and tumor cells for immunotherapy in mouse models of prostatic cancer. Proc Natl Acad Sci U S A 2001;98:12180-5. [Crossref] [PubMed]
- Chudasama V, Maruani A, Caddick S. Recent advances in the construction of antibody-drug conjugates. Nat Chem 2016;8:114-9. [Crossref] [PubMed]


