Thoracic aorta cardiac-cycle related dynamic changes assessed with a 256-slice CT scanner
Introduction
Multidetector computed tomography angiography (CTA) is usually performed for the evaluation of thoracic aneurysms prior to endovascular repair using volumetric ECG-gated acquisition scans in order to obtain images without motion artifacts. Nonetheless, image reconstruction is commonly made using a mid diastolic window. A number of studies have demonstrated that the combination of cardiac pulsatility and aortic compliance leads to conformational changes during the cardiac cycle, both in healthy and diseased vessels (1-3). Indeed, cardiac cycle-related dynamic changes can be measured even in coronary arteries (4). Although these changes have been well documented in the ascending and abdominal aorta, the extent of dynamic conformational changes within the thoracic descending aorta remains poorly understood.
Accordingly, the aim of our study was to attempt to demonstrate the presence of significant differences in aortic size during the cardiac cycle, that might deserve attention given the potential impact for the selection of the type and size of aortic endografts.
Methods
The present was a single-centre, investigator-driven, observational study that aimed to explore whether cardiac-cycle related dynamic changes are associated to significant changes of the descending thoracic aortic diameters. For that purpose, a consecutive series of patients referred to our institution to undergo CTA of the thoracic aorta within the period comprised between March and June 2012 constituted the study population. Inclusion criteria included adult patients (≥18 years), without a history of contrast related allergy, renal failure, or hemodynamic instability. Patients with diffuse aortic atherosclerosis, pacemakers, implantable devices, or previous thoracic endografts were excluded from the analysis. Baseline heart rate or body mass index did not impact the enrollment decision. The institution’s Ethics Committee approved the study protocol, which complies with the Declaration of Helsinki, and written informed consent was obtained from all patients.
CTA acquisition
All studies were acquired with a 256-MDCT scanner (Brilliance ICT, Philips Healthcare, Cleveland, US). A single bolus of 70 mL of iobitridol (Xenetix 350TM, Guerbet, France) was injected through an arm vein at 4 mL/second, followed by a saline chaser. A bolus tracking technique was used to synchronize the arrival of contrast at the thoracic aorta with the start of acquisition. Scanning parameters were as follows: tube voltage 100 kV; tube current 200 mAs; slice width 1 mm; reconstruction interval 0.5 mm. ECG-gating was performed in all cases, and images were reconstructed from 0% to 90% of the R-R interval with 10% stepwise increments; being 30-40% and 70-78% commonly selected as systolic and mid-diastolic phases, respectively. Furthermore, ungated images were retrospectively reconstructed and measurements were performed accordingly.
CTA analysis
Images were transferred to a dedicated CT workstation (Brilliance Workspace, Philips Healthcare, Cleveland, Ohio, USA), and analyses were performed by an experienced level 3—certified coronary CTA radiologist using dedicated software (Advanced Vessel Analysis, Philips Healthcare). Measurements were performed at the motionless systolic (30-40% of the R-R interval) and diastolic (70-78% of the R-R interval) phases. Two orthogonal imaging planes (maximal and minimal diameters) and the lumen area based on manual planimetry were obtained at three different levels of the descending thoracic aorta, using the distance from the left subclavian artery as proximal landmark: 10, 40, and 80 mm distance; and at 40 mm distance of the aortic valve (ascending aorta). Anatomical landmarks such as mild calcifications were used in addition to the distance from the left subclavian artery to achieve accurate co-localization of the region of interest.
Statistics
Discrete variables are presented as counts and percentages. Continuous variables are presented as means ± SD. Comparisons between groups were performed using paired sample Student’s t-test. A two-sided P-value of less than 0.05 indicated statistical significance. Statistical analyses were performed with the use of SPSS software, version 13.0 (Chicago, IL, USA).
Results
A consecutive series of 30 patients referred to undergo CTA of the thoracic aorta constituted the study population. The mean age was 58.9±15.7 years and 16 (53%) patients were male.
The mean ungated maximal aortic diameter was 31.9±5.6 mm at 40 mm distance from the aortic annulus; and 25.3±3.4, 25.7±4.3, and 24.2±4.5 mm at 10, 40, and 80 mm distance from the left subclavian artery, respectively.
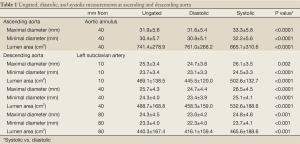
All systolic measurements at the ascending aorta were significantly larger than diastolic measurements (Table 1, Figure 1). Likewise, descending aorta measurements at 10, 40, and 80 mm distance from the left subclavian artery were all significantly larger within the systolic window (Table 1).
Full Table
Measurements of the maximal diameter were systematically larger than the minimal diameters among all aortic positions including ungated, systolic, and diastolic measurements (P<0.001 for all comparisons except for diastolic measurements of the ascending aorta at 40 mm from the annulus; P=0.02). Moreover, 10 (33%) patients had ≥3 mm difference (the established cutoff point to define an aortic oval shape) in diameter between maximal and minimal measurements (5). The mean effective dose was 6.8±1.0 mSv.
Discussion
Our main findings can be summarized as follows: (I) the thoracic descending aorta undergoes significant conformational changes during the cardiac cycle; (II) rather than a circular shape, the thoracic descending aorta has a relatively oval shape.
Regardless of the distance from the left subclavian artery, we demonstrated significantly larger measurements in systolic reconstructions. These results, which are in line with previous reports that established the presence of considerable conformational dynamic changes at both the ascending and abdominal aorta, should support the routine use of electrocardiographic gating for CTA studies performed prior to endovascular repair of the thoracic aorta.
It should be stressed that we performed measurements using orthogonal views, which has been established as the best approach to achieve reproducible measurements of the thoracic aorta. Indeed, previous studies that evaluated the dynamic changes in the aortic root failed to show significant differences, being this lately attributed to the fact that these studies reported measurements using coronal and single oblique sagital views (5,6).
It has recently been emphasized that the aortic valve annulus has an oval shape, being this clinically relevant for the selection of aortic valves in the context of transluminal aortic valve replacement. Furthermore, it has been described that aside from the aortic annulus, the aortic root as well as the sinotubular junction also have an oval shape (1). It is noteworthy that rather than a circular shape, we found significant differences between maximal and minimal diameters irrespective from the distance from the left subclavian artery, suggesting a fairly oval anatomic shape. Whether these findings have potential clinical implications regarding the selection of aortic endografts remains outside the scopus of our investigation and deserves further exploration in appropriately designed prospective studies.
Limitations
A number of limitations should be recognized. The relatively small sample size might potentially lead to selection bias. Furthermore, we decided not to include patients with diffuse aortic disease in order to attain a homogeneous sample and to simulate the anatomy of the proximal aneurismal neck, although we acknowledge that this might represent a limitation of the study.
Conclusions
The main finding of our pilot investigation was that the thoracic descending aorta undergoes significant conformational changes during the cardiac cycle, irrespective from the distance from the left subclavian artery.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- de Heer LM, Budde RP, Mali WP, et al. Aortic root dimension changes during systole and diastole: evaluation with ECG-gated multidetector row computed tomography. Int J Cardiovasc Imaging 2011;27:1195-204. [PubMed]
- Teutelink A, Muhs BE, Vincken KL, et al. Use of dynamic computed tomography to evaluate pre- and postoperative aortic changes in AAA patients undergoing endovascular aneurysm repair. J Endovasc Ther 2007;14:44-9. [PubMed]
- van Herwaarden JA, Bartels LW, Muhs BE, et al. Dynamic magnetic resonance angiography of the aneurysm neck: conformational changes during the cardiac cycle with possible consequences for endograft sizing and future design. J Vasc Surg 2006;44:22-8. [PubMed]
- Rodriguez-Granillo GA, Agostoni P, García-García HM, et al. In-vivo, cardiac-cycle related intimal displacement of coronary plaques assessed by 3-D ECG-gated intravascular ultrasound: exploring its correlate with tissue deformability identified by palpography. Int J Cardiovasc Imaging 2006;22:147-52. [PubMed]
- Tops LF, Wood DA, Delgado V, et al. Noninvasive evaluation of the aortic root with multislice computed tomography implications for transcatheter aortic valve replacement. JACC Cardiovasc Imaging 2008;1:321-30. [PubMed]
- Wood DA, Tops LF, Mayo JR, et al. Role of multislice computed tomography in transcatheter aortic valve replacement. Am J Cardiol 2009;103:1295-301. [PubMed]


