
Intraluminal fibrous cord of the aorta in the differential diagnosis of aortic dissection
Intimal flap is thought to be the characteristic imaging sign of aortic dissection. Nowadays aortic computed tomography angiography (CTA) plays a pivotal role in confirmation of diagnosis and the classification and localization of intimal tears. However, other anomalies resembling the intimal flap should also be considered. We present a rare case with an intraluminal fibrous cord in the distal segment of ascending aorta, mimicking the flap of aortic dissection.
Case presentation
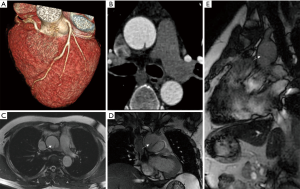
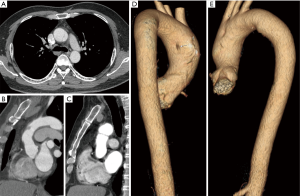
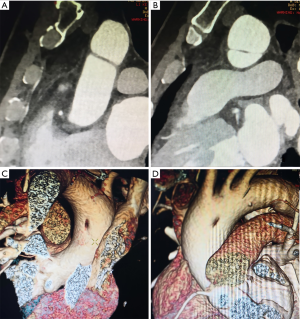
A 47-year-old man with diabetes mellitus but no obvious symptoms of coronary artery disease (CAD) underwent a routine health examination, which contained a coronary CTA examination. A suspicious intraluminal line was found (Figure 1A,B). Even though the coronary CTA images did not include the entire structure, an aortic dissection was suspected. Considering repeat radiation and contrast agent exposure, a magnetic resonance imaging (MRI) scan was recommended. MR showed a low signal intraluminal line in the distal segment of ascending aorta. The ends of the line connected tightly to inner wall of the aorta. Using fast imaging employing steady-state acquisition (FIESTA) sequences, cine images showed absence of movement or shift of the intraluminal line during the entire cardiac cycle, and equal blood flow signal in the “half cavities” adjacent to the cord (Figure 1C,D,E). One month later, the patient requested an aorta CTA examination. The aorta CTA showed stable appearance of the intraluminal fibrous cord in the distal segment of ascending aorta (Figure 2A). The multiplanar reconstruction showed that the fibrous cord obliquely connected the anterior and posterior wall of ascending aorta, and bisected the aorta lumen (Figure 2B,C). The fibrous cord was about 24.5 mm in length and 2 mm in diameter. Volume rendering (VR) showed each end of the fibrous cord had tentiform attachments to the aorta inner wall (Figure 2D,E). The patient did not consider surgical options. The cardiovascular surgeon suggested an annual routine vascular examination in order to monitor the change of the intraluminal fibrous cord. The radiologist advised follow-up imaging with CTA or MRI in a year. CTA or MRI were done once a year over 5 years’ follow-up period with no significant lesion change (Figure 3).
Discussion
This patient presented with an asymptomatic intraluminal fibrous cord mimicking the flap of an aortic dissection in the distal segment of ascending aorta. In the differential diagnosis, chronic residuals of a focal aortic dissection/injury and congenital variants of the aorta were considered (1). In contrast to the described case, residuals of chronic aortic dissection flaps are typically irregular thickened, nodular, or calcified.
Therefore, we favored the hypothesis that the fibrous cord was consistent with a congenital variant. Aortic development begins during the 3rd week of gestation. It is a complex process, which lends itself to a variety of congenital variants and pathologic anomalies. Six paired primitive, or pharyngeal, aortic arches develop between the ventral and dorsal aortae (2). The primitive 4th embryologic arches contribute to the definitive adult aortic arch, and the primitive 6th arches contribute to the ductus arteriosus and central pulmonary arteries. The 5th primitive arches, which is just between the 4th and 6th embryologic arches, typically do not form, or they form incompletely and then regress. Persistent 5th aortic arch (PFAA) is a rare congenital anomaly of the aortic arch, associated with tetralogy of Fallot, transposition of the great arteries, truncus arteriosus, and aortic arch coarctation or interruption. PFAA originates from the distal ascending aorta and is distally connected with the aorta arch or descending aorta. In Weinberg type A, PFAA forms an additional aortic arch parallel to the primary aortic arch (3). Naimo et al. reported a PFAA case presenting as a double-lumen aortic arch in association with a ventricular septal defect. They explained that the double-lumen aortic arch was due to the PFAA associated with interruption of the 4th aortic arch (4). One possible explanation for the current case might be that the PFAA was incompletely formed and then rapidly regressed, which was inadequate to form a double cavity aorta or other aorta-pulmonary artery shunt deformity, but a remnant of intraluminal fibrous cord in the ascending aorta-aortic arch transitional segment. Certainly, a definitive pathological diagnosis of this lesion would require surgical excision.
Aortic CTA has become the most widely used diagnostic modality for a large number of clinical situations, with many advantages including less-invasive nature, wide availability, rapid acquisition, sub-millimeter spatial resolution and high value in guiding patient management. However, incidental findings may lead to diagnostic errors (5). We present a very rare case with an intraluminal fibrous cord of ascending aorta, mimicking the flap of aortic dissection. In the current case, multi-modality imaging could provide different aspects of information (CTA for the shape, MR for blood flow of double cavities), and advanced post processing techniques, such as multiplanar reconstruction and VR, provided a more comprehensive view of the whole lesion. Further, CTA/MRI follow-up imaging allowed documentation of stability. Our case demonstrates that multi-modality imaging and post-processing techniques play an important role in interpretation of the small structures and differential diagnosis of incidental findings. Recognition of these anomalies and differentiation from, e.g., aortic dissection, has clinical significance, and avoid more invasive examination or unnecessary interventions.
Acknowledgments
Funding: This work was supported by China National Natural Science Foundation (grant No. 81301217 and 81301202), Tianjin Research Program of Application Foundation and Advanced Technology (grant No. 18JCYBJC25100 and 14JCZDJC57000).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Hanneman K, Newman B, Chan F. Congenital Variants and Anomalies of the Aortic Arch. Radiographics 2017;37:32-51. [Crossref] [PubMed]
- Nishi K, Inamura N, Marutani S, et al. Rare basis of patent ductus arteriosus: Persistence of the fifth aortic arch. Pediatr Int 2017;59:1091-3. [Crossref] [PubMed]
- Cetrano E, Polito A, Trezzi M, et al. Neonatal Repair of Persistent Fifth Aortic Arch Coarctation and Interrupted Fourth Aortic Arch. Ann Thorac Surg 2017;103:e475-7. [Crossref] [PubMed]
- Naimo PS, Vazquez-Alvarez Mdel C, d'Udekem Y, et al. Double-Lumen Aortic Arch: Persistence of the Fifth Aortic Arch. Ann Thorac Surg 2016;101:e155-6. [Crossref] [PubMed]
- Baliyan V, Verdini D, Meyersohn NM. Noninvasive aortic imaging. Cardiovasc Diagn Ther 2018;8:S3-18. [Crossref] [PubMed]


