Local innovation for improving primary care cardiology in resource-limited African settings: an insight on the Cardio Pad® project in Cameroon
Commentary
Cardiovascular disease (CVD) is a leading cause of death globally. Furthermore, CVD disproportionally affects low- and middle-income countries (LMIC), where about 80% of deaths from CVD around the world are recorded (1). Even in countries within Africa, CVD and non-communicable diseases (NCDs) in general are set to overtake HIV infection, malaria and tuberculosis as the leading cause of death. Although CVD is increasingly receiving attention across Africa, countries in this region are still to develop economically, confronted with the high burden of infectious diseases, and are still ill-prepared to cope with the rising epidemic of CVD. Major challenges include the inefficient and inadequate health facilities, poor health system organization and lack of qualified health workforce to meet increasing healthcare demands for CVD (2).
To overcome the current health workforce shortage in African countries, some strategies such as the task shifting and telehealth have been suggested as means for rapidly increasing access to prevention and curative services for emerging CVD and NCDs (3,4). Task shifting involves a process of rational delegation whereby tasks are moved, where appropriate, from highly qualified health workers (for example specialists doctors) to health workers with shorter training and lower qualifications (for example nurses) in order to make more efficient use of the available human resources for health. Task shifting has been shown to be a key element in successful health interventions in Africa such as the scaling up of HIV-programs and the management of hypertension and diabetes in some settings (5). However, task shifting alone is not expected to resolve the health workforce crisis, and should therefore be implemented alongside complementary strategies as advocated by the World Health Organization (WHO) (3).
With 60% of the African population living in rural areas and considering the extreme shortage of health professionals in rural Africa, telehealth is seen as a mean for improving equitable and affordable access to healthcare services across countries in Africa, particularly for the emerging CVDs (6). Indeed, telemedicine can significantly contribute to the implementation of primary care prevention and control interventions for CVD that have been proven to be highly effective (2). The WHO has defined telemedicine as ‘The delivery of health care services, where distance is a critical factor, by all health care professionals using information and communication technologies for the exchange of valid information for diagnosis, treatment and prevention of disease and injuries, research and evaluation, and for the continuing education of health care providers, all in the interests of advancing the health of individuals and their communities’ (3). While telemedicine is widely available in developed countries using complex technology, its uptake in Africa remains poor (6).
Several telemedicine projects have been initiated within the African continent, some of which have been successful, particularly in the field of tele-education. Examples of such successful telemedicine initiatives include the ‘Réseau en Afrique Francophone pour la Télémédecine’ (RAFT) based at the Geneva University Hospitals in Switzerland and which is active in 15 West African countries; the iPath, an international open source web-based platform for store-and-forward clinical telemedicine; The African Teledermatology Project which offers free store-and-forward teledermatology services; and the IKON project, a teleradiology project in Mali (6). However, most of telemedicine initiatives in sub-Saharan Africa have not led to sustained clinical telemedicine services. The second survey of the WHO’s Global Observatory for eHealth (GOe) has suggested that motivations and challenges to the adoption of telehealth differ between developed and developing countries (4). As such lessons learned from implementing telehealth in developed countries cannot be used uncritically to inform the uptake of teleheath in developing countries. Context-relevant strategies are therefore needed. The Cardio Pad® initiative seeks to provide some of these contextually appropriate solutions to improve the application of telemedicine for CVD prevention and control in remote African settings.
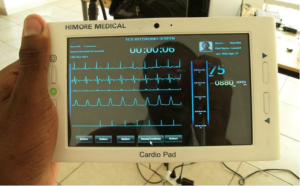
The Cardio Pad® (Figure 1) is the result of dedicated years of efforts by the 26-year-old Cameroon-based engineer Arthur Zang. Arthur started developing this device in 2009 during his engineering training at the Polytechnic High School of the Yaoundé I University, Cameroon. He first designed and developed a system for acquisition, processing and transmission of cardiac signal using the global system for mobile (GSM). This application was tested and the results compared with those of other researchers using the MIT MIB database. The results showed a 98% success in the digital cardiac signal processing (7). A prototype of the Cardio Pad® was available already by 2010, and subsequent efforts have focused on refining the tool, testing it and exploring avenues for mass production and marketing. The efforts of Zang in developing Cardio Pad® have been recognized well beyond Cameroon and Africa at large, with the recent award of the 2014 Rolex Award for Enterprise, for his invention, which is credited to be the African’s first medical computer tablet (8).
The Cardio Pad® is a 7-inch fully touch screen PC tablet with a lithium battery that provides an autonomy for more than seven hours. The motherboard is made up of a dual core processor Texas Instrument OMAP3530 (a ARMV4 processor with a 600 mHz speed and a DSP processor with a 200 mHz speed for digital signal processing), a DDR RAM of 256 MB, an internal memory of 2 GB, a GSM/GPRS for data transmission, a SIM card connector, a GPS module, a Bluetooth module to read patient data, a Wi-Fi module, two internal speakers and a microphone port. The Cardio Pad® uses a personalized operating system (OS) developed from the Microsoft® Windows® CE 5.0 32-bit OS. The Cardio Pad® has four main applications: (I) electrocardiograms (ECG) PAdExam to perform 3, 6 or 12 leads ECG; (II) ECG PadScope which performs real-time ECG monitoring along with other vital parameters such as oximetry, respiratory rate, blood pressure, temperature, and provides an alert in case of any anomaly; (III) ECG PadAnalyse which presents patient’s data to the cardiologist and assist him interpreting the data and providing computer-assigned diagnosis; (IV) Pad D-Transfer to transfer the data from one Cardio Pad® to another or to the National Cardiography Data Remote Center. Other applications such as the cardiology encyclopedia are being developed.
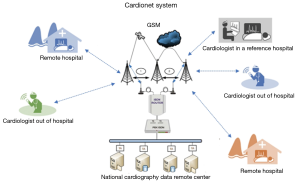
When used as an ECG device, wireless electrodes and a sensor are attached to the patient, which then send signal to the tablet via Bluetooth connection. Using an appropriate software, the signal is treated and stored in a SQL server compact. The ECG with the medical data of a patient in a remote underserved area for example is transferred via mobile phone connection to the National Cardiography Data Remote Center where data are stored. Data are then sent to the cardiologist/specialist on his Cardio Pad®; who after interpretation, sends the report with suggestions for case management to the patient’s care provider in the remote area (Figure 2).
The Cardio Pad® has a number of advantages in terms of cost, ease of use, autonomy and reduced technology requirements. The anticipated marketing price of about $2,000 suggests that Cardio Pad will even be cheaper than existing portable electrocardiographs (8). The built-in battery provides an autonomy for more than seven hours which will be very useful in remote villages where there is no or only limited power supply. The Cardio Pad® will be used in all areas served with mobile phone network.
No implementation study has been conducted to assess if the introduction of Cardio Pad® in clinical setting will affect the behavior of healthcare provider and decision making, and in the long run affect the outcomes of care. However, requirements and challenges of conducting such studies shouldn’t be a deterrent to the uptake of the device in routine settings in remote areas, particularly when the baseline situation is zero; that is when there is no access to the basic ECG testing in remote areas. The impact of tele-ECG, a key functionality of Cardio Pad®, has been well demonstrated in western countries, where its implementation has significantly improved timely management of acute coronary syndromes by para-medical staff (4). A recent study conducted in Cameroon (the birth country of Cardio Pad®), the TELEMED-CAM study, evaluated the efficiency of telemanagement of hypertension in rural health facilities. The study involved a telemedicine center based at a reference hospital in the capital city of Cameroon, the Yaoundé General Hospital which has five cardiologists available, and 30 remote rural health centers led by general practitioners and nurses. The study showed that intervention package comprising tele-support to general practitioners and nurses was effective in improving the management of hypertension and outcomes of care (9). Similarly to the TELEMED-CAM study, a pilot tele-cardiology project with the Cardio Pad® is currently underway in some rural health facilities as a first step for further nationwide implementation.
While most of the current telemedicine clinical services on the African continent receive most expertise from developed countries, the Cardio Pad®, a local invention by a locally trained expert demonstrates how many innovative solutions to combat CVD and other health issues could and should be developed locally in Africa. While waiting for scientific evidence of the benefit of the Cardio Pad® in the targeted communities, there is an increasing hope that telemedicine will contribute to the intensive efforts to strengthen primary care health system for CVD in African countries.
Acknowledgements
We are grateful to Arthur Zang for providing the technical data for his device.
Disclosure: The authors declare no conflict of interest.
References
- World Health Organization. eds. Global Status Report on Non-communicable Diseases 2010. Geneva: World Health Organization, 2010.
- Peck R, Mghamba J, Vanobberghen F, et al. Preparedness of Tanzanian health facilities for outpatient primary care of hypertension and diabetes: a cross-sectional survey. Lancet Glob Health 2014;2:e285-92. [PubMed]
- World Health Organization’s HIV/AIDS Programme. Strengthening health service to fight HIV/AIDS, Taking stock: Task shifting to tackle health worker shortages. Available online: http://www.who.int/healthsystems/task_shifting_booklet.pdf
- World Health Organization. Telemedicine: Opportunities and Developments in Member States: Report on the Second Global Survey on eHealth 2009 (Global Observatory for eHealth Series, Volume 2). Healthc Inform Res 2012;18:153-5.
- Lekoubou A, Awah P, Fezeu L, et al. Hypertension, diabetes mellitus and task shifting in their management in sub-Saharan Africa. Int J Environ Res Public Health 2010;7:353-63. [PubMed]
- Mars M. Telemedicine and advances in urban and rural healthcare delivery in Africa. Prog Cardiovasc Dis 2013;56:326-35. [PubMed]
- Kingue S, Zang A, Tangha C, et al. The detection of QRS complex in a numerised electrocardiographic signal. Results from 2-lead recording in Cameroon. Cameroon Medical Informatics Congress, 2009.
- Rolex Awards for Enterprise. 2014 Young Laureates-Cardiac telemedicine brings hope in Cameroon. Available online: http://www.rolexawards.com/profiles/young_laureates/zang_arthur/project
- Kingue S, Angandji P, Menanga AP, et al. Efficiency of an intervention package for arterial hypertension comprising telemanagement in a Cameroonian rural setting: The TELEMED-CAM study. Pan Afr Med J 2013;15:153. [PubMed]