Screening echocardiograms in a senior focused value based primary care improves systolic heart failure detection and clinical management
Introduction
Heart failure (HF) affects over 5 million people in the United States (1). This number is projected to increase due to aging population and improvement in survival rates for co-existing medical conditions (2).
Unrecognized left ventricular systolic dysfunction has an estimated prevalence of 3% to 6% (3). Because it often occurs in the absence of known cardiovascular disease, this condition may go unrecognized and undertreated. In randomized trials, individuals with asymptomatic left ventricular systolic dysfunction have high rates of incident HF and death (3).
Current guidelines do not support community-wide screening for asymptomatic left ventricular systolic dysfunction, either with echocardiography or with assays for natriuretic peptides (3-5). Nevertheless, the Affordable Care Act emphasizes the provision of preventive patient-centered care, raising the question of the role that screening echocardiograms may have in risk-stratification and prevention of major adverse cardiovascular events among older adults receiving primary care. The emergence of therapeutic options that can halt the progression HF and reduce cardiovascular related mortality makes this question even more relevant (6,7). In this evolving therapeutic context, identifying asymptomatic heart failure (Stage B) could become a valid preventive strategy.
Therefore, the aim of our study was to evaluate the impact of screening echocardiograms on the identification of Stage B and Stage C systolic HF and on the use of cost-effective therapies among older adults receiving care in a network of clinics across 6 states in the United States.
Methods
Study setting
We conducted the study in Chen Neighborhood Medical Centers (CNMC)/JenCare Neighborhood Medical Center (JCNMC) a fully capitated group network practice in six states. Participating patients are insured through Medicare Advantage Plans. The study was approved by the institutional review board (ID#20140187).
Echocardiogram
We offered an abridged echocardiogram to all CNMC or JCNMC members across 6 states who attended a clinic visit between January 1, 2014 and March 20, 2015. Patients signed informed consent and had the echocardiogram performed according to American Society of Echocardiography guidelines (8). Measurements of LV ejection fraction (EF) were performed using the modified biplane Simpson’s rule as a mean of three cardiac cycles. EF less than 50% was used as a cutoff for abnormal LVEF (LV systolic dysfunction). Diastolic function was classified according to recent recommendations of ASE on diastolic functional evaluation (9). The grading scheme was mild or grade I (impaired relaxation pattern), moderate or grade II (pseudonormalized filling), and severe (restrictive pattern) or grade III (9). Each echocardiogram was reviewed by a board certified cardiologist blinded to the patient’s history. The echocardiogram results were made available to the caring primary care physician within 24 hours.
Study design and study population
We conducted a pre-post analysis of the CNMC population who had the screening echocardiogram during the study period. For our analysis, we focused on older adults with a higher prevalence of HF (10). We defined this group as any adult greater than 60 years of age. We considered the 6 months before the screening echocardiogram as the pre-echococardiogram period and the 6 months after the echocardiogram as the post-echocardiogram period. We diagnosed HF for 6 months before the echocardiogram using ICD-9 code (428.0,1,2,4). The code could be in any position in the medical claims file during the 6 months prior to the screening echocardiogram. These diagnoses were triggered by either a clinically indicated echocardiogram or a hospitalization due to heart failure, The use of ICD-9 code is a valid method of identifying HF with a PPV of >90% (11).
Primary outcome
Our primary outcome for the post-echocardiogram period was incident depressed left ventricular ejection fraction (EF) defined as an EF of <50%. We further dichotomized this outcome into Stage B for patients with depressed EF but without evidence of heart failure symptoms and Stage C for those who had reported them. For the primary outcome we excluded all subjects who already had a diagnosis of heart failure in the pre-echocardiogram period.
Definition of stage B heart failure
A cardiologist (RG) and a internist (AG) evaluated all the primary care or cardiology visits in the electronic medical record of the patients who had a depressed Ejection Fraction in the screening echocardiogram. We reviewed all notes in the pre and post echocardiogram periods. We defined Stage B HF as the absence of dyspnea, orthopnea, paroxysmal nocturnal dyspnea or pedal edema 3 months before or after the date of the echocardiogram (12).
Secondary outcomes
We evaluated two secondary outcomes.
Use of evidence based medications: We collected the prescription of generic cost-effective HF evidence based medications through two strategies: identifying NDC codes from pharmacy claims data and identifying medication names from the electronic medical record. We collected the use of beta-blocker, ace-inhibitor and diuretic due to their beneficial impact on HF symptoms and outcome. Although aspirin and statins due not influence HF outcomes, we collected their use due to their role on outcomes among patients who might have concomitant CAD. We defined a medication as prescribed if a medication in each of the specified classes was present either in claims data or in the medication lists 6 months before or 6 months after the echocardiogram. We calculated the absolute increase in medication use (13). To further characterize the use of medications we report the use according to the absence or presence of HF symptoms (Stages B or C respectively) and to an EF lower or higher than 40% in the screening echocardiogram. The guidelines recommend the use of certain medications such as Ace inhibitors for patients with HF lower than 40%. In patients with normal ejection fraction there was no change in the use of ace-inhibitor or beta-blocker (P>0.05).
Intermediate clinical outcomes: We collected from the electronic medical record all the values for body mass index, systolic blood pressure, diastolic blood pressure and heart rate before and after the echocardiogram. We also collected laboratory data on low density lipoprotein (LDL) from the electronic medical record. For the pre-echocardiogram analyses, we used the intermediate clinical outcomes measured immediately before having the echocardiogram. For the post-echocardiogram analyses we used the last intermediate clinical outcome 6 months after the echocardiogram.
Other covariates
Using the electronic medical record, we collected demographic information and presence of comorbidities. We calculated the Charlson score as a measure of disease burden. This is a validated method to assess co-morbidity status (14).
We also divided the population by the number of CV risk factors. We considered risk factors as hypertension, diabetes, hyperlipidemia and obesity and this data was obtained from the medical record using standard definitions (15).
Statistical analyses
We evaluated the distribution of the continuous variables using measures of central tendency and skewedness. To compare the number of prevalent and incident cases of Heart Failure we used chi-square. We report the number and proportion of subjects with newly diagnosed Stage B Heart Failure.
To determine predictors of having incident Stage B and Stage C heart failure we used logistic regression to calculate the odds ratio (OR) and the corresponding 95% CI.
To test pre-post intervention differences in heart rate, LDL cholesterol and blood pressure we used paired t-test. To account for the within subject correlation of our measures and the effect of time, we used linear mixed regression models which also included categorical classification variables to index time intervals pre and post intervention. As this was a pre-post comparison on the same subjects, we did not adjust for traditional covariates such as gender or race.
To test pre-post use of evidence based medications we used Chi-Square to compare the proportion of subjects using each class of medications in the entire cohort and stratified by Heart Failure stage and by the EF cutpoint of 40%.
To evaluate the robustness of our findings we conducted two sensitivity analyses. The first analysis stratified the prevalence of heart failure by the number of days between the date of the echocardiogram and the enrollment in CNMC to assure that patients newly enrolled did not join with a diagnosis of HF. Our stratifying variable was the median time. The second analysis stratified the prevalence of HF by the number of cardiovascular risk factors.
The fitness of the data was assessed using the deviance ratio. Analyses were performed using STATA (College Station, Texas), and all significance tests were two-tailed.
Results
Baseline characteristics of the sample
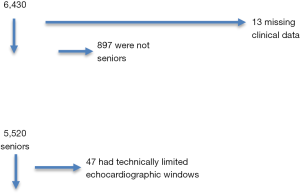
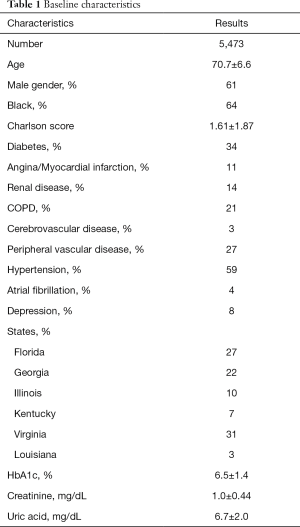
Figure 1 reports the flow-chart of the inclusion of older adults in the study. A total of 6,430 had a screening echocardiogram. We excluded 13 cases due to missing data, 897 who were younger than 60 years of age and 47 due to a technically limited echocardiogram. Table 1 reports the baseline characteristics of the included patients. We included 5,473 subjects with a mean age of 70.7±6.6. Of those, 61% were males and 64% were Black. The mean Charlson score was 1.61±1.87. The majority of the patients resided in Virginia (31%) and Florida (27%).
Full table
Incidence of systolic heart failure after screening echocardiogram
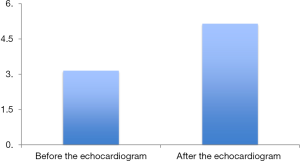
Figure 2 reports the change in the number of HF cases before and after the screening echocardiogram. The screening echocardiogram diagnosed 292 new subjects with a depressed ejection fraction of less than 50%. This represented 5.34%; 95% CI (4.5–5.7) of the population. Of those, 239 out of 292 subjects (82%) had no symptoms of HF and were classified as newly diagnosed Stage B HF and 53 had symptoms and (18%) were diagnosed with Stage C HF. Pedal edema was the most common symptom among the newly diagnosed Stage C. Overall, 4.36% of the entire sample of 5,473 subjects was diagnosed with Stage B HF.
Predictors of incident heart failure
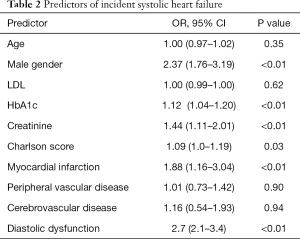
In univariate analysis age, male gender, Charlson score and creatinine were predictors of having a new diagnosis of depressed ejection fraction. Table 2 reports multivariate predictors of incident HF. Significant ORs for a new HF diagnosis were male gender (2.37; 95% CI: 1.76–3.19), HbA1c (1.12; 95% CI: 1.04–1.20), creatinine (1.44; 95% CI: 1.1–2.0), Charlson score (1.09; 95% CI: 1.0–1.19) and myocardial infarction (1.88; 95% CI: 1.16–3.04). Having more cardiovascular risk factor was not a predictor of incident HF.
Full table
Sensitivity analysis
The number of CV risk factors did not increase the detection of incident HF (P=0.13). The mean time between enrollment and the screening echocardiogram was 47 (IQR 22-95) days and the incidence of heart failure in those over and below 47 days was 5%.
Echocardiographic findings
Table 3 reports the results of the screening echocardiogram. Ninety-five percent of subjects had a normal ejection fraction, 2.3% of the entire sample had an EF lower than 40%, 30% had either pseudonormalization or a restrictive pattern as markers of diastolic dysfunction. Only 11% had normal diastolic function.
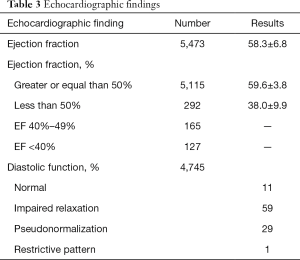
Full table
Effect of the screening echocardiogram on the use of medications
When comparing the use of medications before and after the screening echocardiogram in the entire cohort, we found that the use of ace-inhibitors increased after the echocardiogram by 17%, beta blockers by 10%, aspirin by 12%, statin by 15% and diuretic by 12% (all P<0.01). When evaluating the use of ace inhibitors or angiotensin receptor blocker and beta blockers according to HF stage and ejection fraction we found that among subjects with Stage B HF (Figure 3A) the percentage of use achieved for both classes of medications was slightly higher among those with EF <40%. Among subjects with Stage C (Figure 3B) the overall use was higher when compared to Stage B.
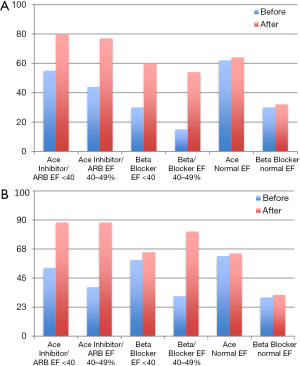
The use of statins before and after the screening echocardiogram among subjects with an LDL higher than 100 mg/dl increased from 45% to 66%.
Effect of the screening echocardiogram on intermediate clinical outcomes
Table 4 reports the changes in intermediate clinical outcomes before and after the screening echocardiogram. Significant decreases were a drop of in 4 mmHg of systolic blood pressure and 8 mg/dL decrease in LDL.
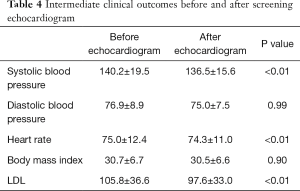
Full table
Discussion
Our study found that among older adults seeking care at a capitated network of multi-specialty medical centers across 6 states, a screening echocardiogram identified a significant number of new cases of HF and 82% of those newly diagnosed patients in our study were found to be Stage B, a group that is very difficult to identify. We also found that among those newly diagnosed, the use of medications for HF, blood pressure, or cholesterol management increased and clinical markers improved after the screening echocardiogram.
Our strengths are a large sample size, the use of the screening echocardiogram for all consecutive patients across 6 states, and the diversity of the sample.
Our study has several limitations. First, we did not have a control group and this limits our ability to evaluate if the detection of new systolic HF led to the change in management or if this occurred as a function of time under the care of CNMC. The fact that we found differences in how medications were used depending on the heart failure stage or the ejection fraction measure suggests the echo informed the use of ace inhibitors and beta-blockers. Second, we only had a single echocardiogram evaluator which could introduce bias. Third, it is difficult to define HF symptoms in older adults because other comorbidities can cause similar symptoms to HF. Fourth, the generalizability of the study is limited to the population we serve which includes elderly Medicare mostly minorities
In population-based surveys, 3% to 5% of the adult population exhibits evidence for ventricular dysfunction likely to progress (16,17). Therapy introduced during this phase can delay or prevent the occurrence of symptomatic HF (7,18). Thus, strategies for the early identification of structural heart disease among undiagnosed individuals could allow the timely initiation of disease-modifying treatment. Due to the low prevalence of structural heart disease among the general population, echocardiography has traditionally not been considered justified in low-risk individuals, with the exception of individuals with a family history of sudden death or hereditary diseases affecting the heart or the great vessels (4). In 2011 several cardiovascular societies evaluated the appropriateness of the use of echocardiography as a diagnostic test in different cardiovascular conditions and labeled the routine surveillance of heart failure in asymptomatic individuals as inappropriate (19).
Two studies have evaluated if screening echocardiograms are useful. Lindekleiv et al. (20) conducted a population-based study where 6,861 middle-aged patients in Norway were randomly assigned to a screening echocardiogram or usual care. The study found 249 patients with new cardiac pathology mostly represented by valvular disease but did find a mortality benefit among those screened. However, this study was population based, included patients aged 25–85 and only 1% had ventricular dysfunction, limiting the generalizability of the results for older adults attending primary care in the US.
The second study, Carerj et al. (5) performed a screening echocardiogram to 1,097 middle age asymptomatic subjects in Italy, classified them according to their cardiovascular risk factor burden and followed them for 2.5 years to determine cardiovascular outcomes. The study found a prevalence of systolic HF that ranged from 8%-15% with increasing risk factor burden and that this new systolic heart failure was predictive of cardiovascular outcomes. The authors concluded that their results supported the performance of screening echocardiograms among patients with 2 or more cardiovascular risk factors. In our study, the screening echocardiogram also yielded a significant number of new HF diagnosis among patients regularly followed in a multi-specialty clinic. However, in contrast, we did not find a yield difference by increasing number of risk factors or other predictors that could translate into a targeted approach. This could be related to the fact that our sample included exclusively older adults who may have a higher pre-test probability than the middle aged population included in the study by Carerj.
Our results suggest that the screening echocardiogram improves the use of cardiovascular medications which in turn may lead to better blood pressure control. This is important in light of SPRINT trial results (21) and accounts for a greater proportion of symptomatic HF in African American and elderly persons and that optimal control of blood pressure reduces HF risk. Interestingly, the initiation of ace inhibitor and beta-blocker therapy post screening echocardiogram seemed to be informed by the EF cut point of 40% more frequently among the Stage B subjects. The initiation of a beta-blocker among symptomatic subjects was less common for those with an EF of less than 40%. At the same time, the echocardiogram seemed to influence the use of medications since we did not see this change in patients with normal ejection fraction.
Only a small proportion of elderly had normal diastolic function, this was expected with the stiffening of the ventricle seen with age and the percentage of patients who cardiovascular risk factors. Our results also found that diastolic dysfunction is a predictor of developing heart failure.
Our results do not support a targeting strategy to select those who would benefit most from the screening echocardiogram as the diagnosed patients were mostly asymptomatic or had non-specific symptoms and had a wide range of burden of cardiovascular risk factors. Even though, our multivariate model identified several significant predictors like gender, creatinine, diabetes and comorbidities, applying this would miss several patients that could benefit from a screening echocardiogram.
Brain natriuretic peptide has been used recently for the screening of heart failure. A study by Hamagawa et al. (22) found that in 393 elderly Japanese rural patients without a diagnosis of heart failure, 7 had some form of systolic dysfunction and the remaining had diastolic dysfunction. This study is limited by the lack of the use of the gold standard in all subjects but certainly suggests an interesting comparison between echocardiography and pro-BNP.
In conclusion, screening echocardiograms among older adults attending capitated multi-specialty medical centers identifies a significant number of patients with stage B Heart Failure. Clinicians who care for elderly patients are often faced with a dilemma when called upon to differentiate between symptoms that could be attributed to cardiac disease, pulmonary disease or simply deconditioning of the patient. This new finding may have led to the improved use of evidence based therapies and control of systolic blood pressure and cholesterol. Future studies should evaluate the effect of screening echocardiograms on cardiovascular outcomes and costs among older adults seeking preventive primary care in the United States.
Acknowledgements
The authors thank Elissa Dawkins and Jorge Alfonso, MD. The study was unfunded.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the institutional review board (ID: 20140187), and written informed consent was obtained from all patients.
References
- Go AS, Mozaffarian D, Roger VL, et al. Executive summary: heart disease and stroke statistics--2014 update: a report from the American Heart Association. Circulation 2014;129:399-410. [Crossref] [PubMed]
- Heidenreich PA, Trogdon JG, Khavjou OA, et al. Forecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart Association. Circulation 2011;123:933-44. [Crossref] [PubMed]
- Wang TJ, Levy D, Benjamin EJ, et al. The epidemiology of "asymptomatic" left ventricular systolic dysfunction: implications for screening. Ann Intern Med 2003;138:907-16. [Crossref] [PubMed]
- Cheitlin MD, Armstrong WF, Aurigemma GP, et al. ACC/AHA/ASE 2003 guideline update for the clinical application of echocardiography: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASE Committee to Update the 1997 Guidelines for the Clinical Application of Echocardiography). Circulation 2003;108:1146-62. [Crossref] [PubMed]
- Carerj S, La Carrubba S, Antonini-Canterin F, et al. The incremental prognostic value of echocardiography in asymptomatic stage a heart failure. J Am Soc Echocardiogr 2010;23:1025-34. [Crossref] [PubMed]
- Claggett B, Packer M, McMurray JJ, et al. Estimating the Long-Term Treatment Benefits of Sacubitril-Valsartan. N Engl J Med 2015;373:2289-90. [Crossref] [PubMed]
- Packer M, McMurray JJ, Desai AS, et al. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation 2015;131:54-61. [Crossref] [PubMed]
- Gottdiener JS, Bednarz J, Devereux R, et al. American Society of Echocardiography recommendations for use of echocardiography in clinical trials. J Am Soc Echocardiogr 2004;17:1086-119. [PubMed]
- Wan SH, Vogel MW, Chen HH. Pre-clinical diastolic dysfunction. J Am Coll Cardiol 2014;63:407-16. [Crossref] [PubMed]
- Daamen MA, Hamers JP, Gorgels AP, et al. Heart failure in nursing home residents; a cross-sectional study to determine the prevalence and clinical characteristics. BMC Geriatr 2015;15:167. [Crossref] [PubMed]
- Saczynski JS, Andrade SE, Harrold LR, et al. A systematic review of validated methods for identifying heart failure using administrative data. Pharmacoepidemiol Drug Saf 2012;21 Suppl 1:129-40. [Crossref] [PubMed]
- Whittaker BD, Soine LA, Errico KM. Patient and process factors associated with all-cause 30-day readmission among patients with heart failure. J Am Assoc Nurse Pract 2015;27:105-13. [Crossref] [PubMed]
- McMurray JJ. Clinical practice. Systolic heart failure. N Engl J Med 2010;362:228-38. [Crossref] [PubMed]
- Sharabiani MT, Aylin P, Bottle A. Systematic review of comorbidity indices for administrative data. Med Care 2012;50:1109-18. [Crossref] [PubMed]
- Grundy SM, Pasternak R, Greenland P, et al. Assessment of cardiovascular risk by use of multiple-risk-factor assessment equations: a statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation 1999;100:1481-92. [Crossref] [PubMed]
- McDonagh TA, Morrison CE, Lawrence A, et al. Symptomatic and asymptomatic left-ventricular systolic dysfunction in an urban population. Lancet 1997;350:829-33. [Crossref] [PubMed]
- Rodeheffer RJ. Epidemiology and screening of asymptomatic left ventricular dysfunction. J Card Fail 2002;8:S253-7. [Crossref] [PubMed]
- Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. The SOLVD Investigattors. N Engl J Med 1992;327:685-91. [Crossref] [PubMed]
- American College of Cardiology Foundation Appropriate Use Criteria Task Force, American Society of Echocardiography, American Heart Association, et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 Appropriate Use Criteria for Echocardiography. A Report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, American Society of Echocardiography, American Heart Association, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Critical Care Medicine, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance American College of Chest Physicians. J Am Soc Echocardiogr 2011;24:229-67. [PubMed]
- Lindekleiv H, Lochen ML, Mathiesen EB, et al. Echocardiographic screening of the general population and long-term survival: a randomized clinical study. JAMA Intern Med 2013;173:1592-8. [Crossref] [PubMed]
- SPRINT Research Group, Wright JT Jr, Williamson JD, et al. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N Engl J Med 2015;373:2103-16. [Crossref] [PubMed]
- Hamagawa K, Kubo T, Nishimura K, et al. N-terminal pro-brain natriuretic peptide is a useful marker to identify latent heart failure patients in older adults in a rural outpatient clinic. Geriatr Gerontol Int 2016. [Epub ahead of print]. [Crossref] [PubMed]