
Trends and effect of atrial fibrillation on inpatient outcomes after transcatheter aortic valve replacement
Introduction
Transcatheter aortic valve replacement (TAVR) has emerged as a less invasive treatment for patients with aortic stenosis at prohibitive, high and intermediate surgical risk (1). Since its inception, better patient selection, better techniques and expertise in TAVR have led to improving patient outcomes (2). As we expand the benefit of TAVR to patients at a lower surgical risk, it is more important than ever to explore the prognostic indicators of patient outcomes after TAVR.
Atrial fibrillation (AF) is common in patients undergoing TAVR (3). AF is also a well-known risk factor for stroke (4), is common in patients with heart failure of valvular origin (5), and is associated with worse outcomes after cardiac procedures such as percutaneous coronary intervention (6) and aortic valve replacement (7). There is conflicting evidence on whether AF impacts various outcomes after TAVR (8,9). It is also unknown if patients undergoing TAVR with AF have seen similar improvements in outcomes over years, as compared to those without AF. Thus, the purpose of this analysis was to assess the effect of AF on inpatient complications, the average length of stay (LOS), and discharge disposition (including inpatient mortality) after TAVR, and to investigate whether improvements in patient outcomes over the years are spread similarly across populations depending on the presence or absence of AF.
Methods
Study design and population
The National Inpatient Sample (NIS) is the largest all-payer database of inpatient hospital stays in the United States and is sponsored by the Agency on Healthcare Research and Quality. It includes data from over 7 million hospitalizations at over 1,000 hospitals each year and represents a 20% stratified random sample of all hospital discharges nationwide (10). The International Classification of Disease, 9th revision, Clinical Modification (ICD-9-CM) diagnostic and procedural codes were used to identify patients.
Hospitalizations of adults ≥50 years old with aortic stenosis (ICD-9-CM 424.1) who had undergone elective TAVR (35.05 and 35.06) from January 1, 2012, to September 30, 2015 (when ICD-10 codes were implemented) were eligible for inclusion. Patients with a history of congenital aortic valve disorders (746.3), rheumatic aortic stenosis (395.0–395.9), or hypertrophic obstructive cardiomyopathy (425.11), who had undergone concomitant procedures involving the coronary vessels (0.61–0.69 and 36.00–36.99), such as percutaneous coronary intervention or coronary artery bypass grafting, who had surgery >2 days after admission (to further limit to elective surgeries), were discharged against medical advice, or had an unknown discharge status were excluded. AF was identified using ICD-9-CM codes 427.31 and 427.32.
The outcomes were inpatient complications, LOS after TAVR, and discharge disposition (including inpatient mortality). Complications of interest included permanent pacemaker placement (PPM) implantation (0.50, and 37.8–37.83), acute kidney injury (584–584.9), transient ischemic attack (TIA)/stroke (344.6–344.61, 431, 433.01, 433.11, 433.21, 433.31, 433.81, 433.91, 434.01, 434.11, 434.91, 435–435.9, and 997.02), acute myocardial infarction (410.01, 410.11, 410.21, 410.31, 410.41, 410.51, 410.61, 410.71, 410.81, 410.91, and 411.1), cardiogenic shock (785.51), cardiac arrest (427.5), bleeding (430, 431, 432–432.9), blood transfusion (99.00–99.09), and vascular complications (39.31, 39.41, 39.49, 39.52, 39.56, 39.57, 39.59, 39.79, 441.0–441.03, 441.1, 441.3, 441.5, 441.6, 414.1–414.19, 443.22, 444–444.9, 447.0, 868.04, 900–904.9, 997.7, 998.2, and 999.2). A composite ‘any complication’ variable was also created. Disposition was categorized as: (I) routine/home health care; (II) transfer to short-term hospital; (III) transfer to skilled nursing facility, intermediate care facility, or other care facility; and (IV) died.
Statistical analysis
Differences in patient and hospital characteristics between hospitalizations of patients with and without AF were compared using Student’s t-tests and Chi-square tests as appropriate. A P value <0.05 was considered statistically significant. Charlson comorbidity index (CCI) was calculated using the Deyo et al. coding scheme (11). Quarterly rates of AF, per 100 TAVR procedures, were estimated using Poisson regression, and changes over time were evaluated using likelihood ratio tests. Additional trend tests were performed to assess changes in in-hospital complications, average LOS after surgery, and discharge disposition over time, stratified by AF. NIS sampling weights were not applied since obtaining national estimates was not a goal of this study.
Multivariable logistic, linear, and generalized logistic regression models were used to estimate the effect of AF on inpatient complications, average LOS after TAVR and discharge disposition, respectively. All models were adjusted for year of TAVR, age, sex, race/ethnicity, CCI, primary insurance, median household income in each patient’s ZIP code, hospital region, hospital type, and hospital size. Both age and CCI were modeled as restricted cubic splines. Because of the low incidence of acute myocardial infarction and cardiogenic shock, adjusted estimates could not be obtained.
All analyses were performed using SAS 9.4 (SAS Inc., Cary, NC, USA).
Results
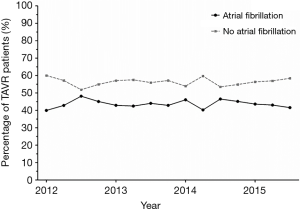
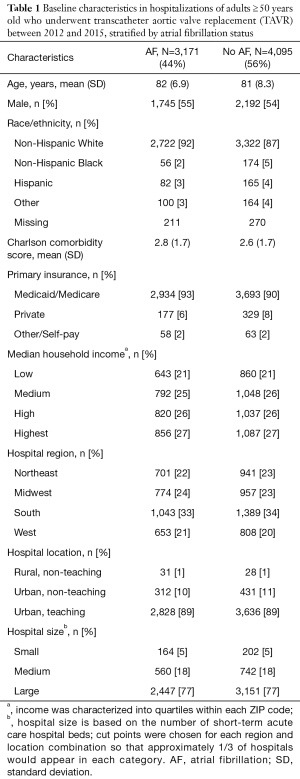
Overall, 7,266 TAVR hospitalizations were included, and 44% had a diagnosis of AF. The proportion of patients with AF undergoing TAVR has remained relatively stable between 2012 and 2015 (P=0.62, Figure 1). CCI also remained stable over this time in both patients with and without AF (P=0.38 and P=0.34, respectively) (data not shown). Patients with AF were more likely to be non-Hispanic white (92% vs. 87%, P<0.0001, Table 1). The vast majority of hospitalizations occurred at urban teaching hospitals (89%), and at large hospitals (77%).
Full table
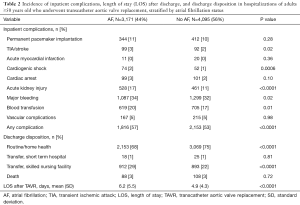
Patients with AF were more likely to have at least one inpatient complication (57% vs. 53%, P<0.0001). Specifically, AF was associated with increased incidences of TIA/stroke (3% versus 2%, P=0.02), cardiogenic shock (2% versus 1%, P=0.0006), acute kidney injury (17% versus 11%, P<0.0001), major bleeding (34% versus 32%, P=0.02), and require a blood transfusion (20% versus 17%, P=0.01) during their hospitalization (Table 2). No significant difference in the incidences of permanent pacemaker implantation, acute myocardial infarction, cardiac arrest, or vascular complications was seen. While the majority of AF and non-AF patients had a routine/home health discharge, patients with AF were more likely to be transferred to a skilled nursing facility (29% versus 22%, P<0.0001). There was no significant difference in inpatient mortality (3% in both groups). Patients with AF also had, on average, longer lengths of stay (6.2 versus 4.9 days, P<0.0001).
Full table
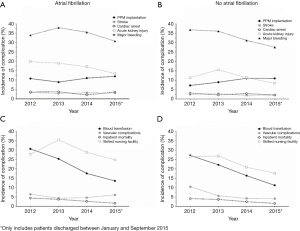
The yearly incidence of PPM implantation, TIA/stroke, cardiac arrest, acute kidney injury, major bleeding, requiring a blood transfusion, vascular complications, transfer to a skilled nursing facility, and inpatient mortality between 2012 and 2015 are depicted in Figure 2. Among AF patients, there was a significant decrease in the incidence of acute kidney injury (20% in 2012, 13% in 2015, P=0.002), requiring a blood transfusion (31% in 2012 versus 14% in 2015, P<0.0001), inpatient mortality (4% in 2012 versus 2% in 2015, P=0.001) and transfer to a skilled nursing facility (28% in 2012 versus 25% in 2015, P=0.009), and the incidence of routine discharge increased significantly (67% in 2012 versus 73% in 2015, P=0.01, Figure 2A,C). The incidence of PPM implantation (P=0.17), TIA/stroke (P=0.98), cardiac arrest (P=0.74), major bleeding (P=0.07), and vascular complications (P=0.64) did not change. In patients without AF, there was also significant decrease in the incidence of acute kidney injury (11% in 2012 versus 8% in 2015, P=0.0002), requiring a blood transfusion (27% in 2012 versus 11% in 2015, P<0.0001), inpatient mortality (4% in 2012 versus 2% in 2015, P=0.003), and transfer to a skilled nursing facility (27% in 2012 versus 18% in 2015, P<0.0001), and an increase in routine discharge (68% in 2012 versus 80% in 2015, P=0.0001), between 2012 and 2015 (Figure 2B,D). However non-AF patients also had a significant decrease in the incidences of major bleeding (37% in 2012 versus 28% in 2015, P<0.0001) and vascular complications (11% in 2012 versus 4% in 2015, P<0.0001). There was also a significant increase in PPM implantation in non-AF patients (7% in 2012 versus 11% in 2015, P=0.01). No changes were seen in the incidence of TIA/stroke (P=0.32) or cardiac arrest (P=0.40).
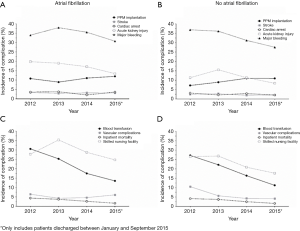
The average LOS decreased, on average, by around 2 days in both patients with and without AF between 2012 and 2015 (7.1 versus 5.3 days and 5.7 versus 4.0 days, respectively, P<0.0001 for both; Figure 3).
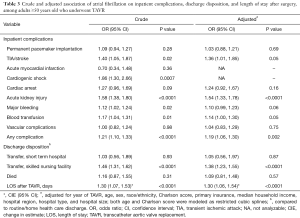
After adjusting for year of TAVR, patient demographics, comorbidities, and hospital characteristics AF was still associated with increased odds of TIA/stroke (OR 1.36, 95% CI: 1.01, 1.85), acute kidney injury (OR 1.54, 95% CI: 1.33, 1.78), and requiring a blood transfusion (OR 1.14, 95% CI: 1.00, 1.30; Table 3). AF was not associated with permanent pacemaker implantation, cardiac arrest, major bleeding or vascular complications. AF was also associated with increased transfer to a skilled nursing facility (OR 1.38, 95% CI: 1.23, 1.55), and an increased LOS [change in estimate (CIE) 1.30, 95% CI: 1.06, 1.54]. AF was not associated with inpatient mortality (OR 1.09, 95% CI: 0.81, 1.48).
Full table
Discussion
Our study found that AF is prevalent among patients undergoing TAVR, and that these patients have increased odds of TIA/stroke, acute kidney injury, requiring a blood transfusion, being transferred to a skilled nursing facility, and increased average LOS. However, there was no difference in inpatient mortality between patients with and without AF. Between 2012 and 2015, the incidence of acute kidney injury, requiring a blood transfusion, inpatient mortality, and average LOS decreased among both patients with and without AF. While major bleeding and vascular complications was consistent in AF patients, the incidence of both of these complications significantly declined in non-AF patients. Similarly, the increase in PPM implantation was only seen in non-AF patients.
We found AF to be prevalent in 44% of patients undergoing TAVR. This is similar to the findings of a previous study among patients with heart failure of valvular origin (5). Additionally, we found that AF patients were more likely to be non-Hispanic white, which has also been previously described (12). Interestingly, while older age is known to be associated with AF, we did not find that in our cohort; however, this may be explained by the fact that TAVR patients were older and sicker during the years we studied here.
As compared to patients without AF, we found that patients with AF were at higher odds of having TIA/stroke, requiring a blood transfusion and acute kidney injury. The association between AF and a higher risk of stroke/TIA is well known (4). Patients with AF were also at higher odds of requiring a blood transfusion. This effect could at least be partly explained by the fact that those with AF are more likely to be on anticoagulants (13). The association of AF with a higher incidence of AKI is also consistent with a previous study (14). Additionally, the association between higher rates of blood transfusion requirement with acute kidney injury post-TAVR has also been noted in previous studies (14,15). Between the study years 2012 to 2015, we found that the incidence of acute kidney injury and requiring a blood transfusion steadily declined, both in those with AF and those without suggesting that the decline in rates of these complications was not dependent on presence or absence of AF.
Patients with AF were also more likely to have an increased LOS after TAVR and be discharged to a skilled nursing facility. The association between pre-existing AF with a longer LOS has been previously investigated (16). In another study of 663 patients undergoing elective TAVR, the absence of AF was predictive of next-day discharge after TAVR (17). There are several possible mechanisms contributing to the increased LOS in patients with AF, including the increased incidence of several complications. Prior research has found that acute kidney injury and postoperative delirium (not studied here) can contribute to longer hospitalizations (15,18). Moreover, longer hospitalizations were previously shown to be associated with functional dependence, which in turn was associated with transfers to a nursing home (19). Although the LOS decreased from 2012 to 2015, it remained higher in patients with AF.
Furthermore, while the odds of having major bleeding or vascular complications were similar in patients with or without AF, there was a decrease in the incidence of major bleeding and vascular complications in the group without AF between 2012 and 2015 but not in the AF group. It is possible that the improving trend in the rates of vascular complications and bleeding was blunted in the AF group due to the effect of anticoagulants. We also found that AF was not associated with PPM implantation, cardiac arrest, or mortality. There is conflicting evidence on the impact of AF on mortality after TAVR. While one study found that AF was not associated with 30-day mortality but was associated with long-term mortality, another analysis found that AF was associated with increased 1-year mortality (9,20). Thus, it is possible that AF only impacts longer-term mortality, which may explain our findings.
Finally, while a similar study utilizing the NIS databases was recently published, our study has several notable differences (21). First, our study spanned across longer study years which may have contributed to the fact that we found a significantly higher rate of TIA/stroke and acute kidney injury in patients with AF. Additionally, as hinted above, we assessed the trends in these complications. Since TAVR has disseminated quickly throughout the US and clinicians have gained expertise in the procedure, as well as the numerous changes to the hardware and procedure itself have been implemented, assessing both the overall effect and trends is of critical importance.
This study has several limitations. The NIS is deidentified database, so we were unable to capture outcomes outside of the index hospitalizations, which only represent a portion of all complications. We are also unable to determine if this study includes repeated observations of patients who underwent more than one TAVR during the study period; however, results were interpreted at the hospitalization-level, not patient-level to account for this. There was also a potential for coding errors and differences in coding practices across the hospitals included in the database. Coding errors could have caused us to underestimate the prevalence of AF, comorbidities, and in-hospital complications. AF misclassification (i.e., labeling a patient as not having AF when they do) would, in expectation, bias results towards the null. Additionally, we suspect that differences in comorbidity and in-hospital complication reporting are random and would not be expected to differ across AF status. This study cannot assess the causal relationship between AF and these outcomes but only an association. While we adjusted for the CCI, we did not have the information needed to calculate surgical risk scores, frailty scores or echocardiographic characteristics, which may have influenced the differences in patient outcomes. Additionally, ICD-9-CM coding prevents us from being able to differentiate between paroxysmal and persistent AF.
Finally, because dates are not attached to diagnosis codes, we had to assume that all patients with AF were diagnosed before they underwent TAVR. While there are wide variations in the reported incidence of new-onset AF in patients undergoing TAVR, an analysis from the SOURCE XT prospective multicenter registry found the prevalence of pre-existing AF was 36% and new-onset AF was only 7%, suggesting that misclassification of pre-existing AF is limited (20). Moreover, in another study, 24-hour ECG monitoring the day before TAVR diagnosed paroxysmal atrial flutter/atrial tachycardia (similar to AF) in 30% of patients who were diagnosed with new-onset atrial flutter/atrial tachycardia after TAVR, suggesting it may have been present before the procedure (22).
Conclusions
After initial concerns about the high incidences of inpatient complications after TAVR, these rates have declined with improvement in TAVR technology, both in those with and without AF. However, AF continues to be associated with higher incidences of inpatient complications, discharge to a skilled nursing facility, and longer average LOS as compared to those without AF. With prevalence in almost half of the patients undergoing TAVR, recognition of the association of AF with adverse outcomes and protocols for closer monitoring of AF patients undergoing TAVR are suggested as we expand TAVR to broader populations.
Acknowledgments
None.
Footnote
Conflicts of Interest: Ms. PD Strassle has received salary support from researchEZ LLC and Dr. S Arora’s spouse has proprietary role in researchEZ LLC. The other authors have no conflicts of interest to declare.
Ethical Statement: Study was reviewed by IRB and rendered exempt.
References
- Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017;70:252-89. [Crossref] [PubMed]
- Auffret V, Lefevre T, Van Belle E, et al. Temporal Trends in Transcatheter Aortic Valve Replacement in France: FRANCE 2 to FRANCE TAVI. J Am Coll Cardiol 2017;70:42-55. [Crossref] [PubMed]
- Holmes DR Jr, Brennan JM, Rumsfeld JS, et al. Clinical outcomes at 1 year following transcatheter aortic valve replacement. Jama 2015;313:1019-28. [Crossref] [PubMed]
- Kannel WB, Wolf PA, Benjamin EJ, et al. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol 1998;82:2n-9n. [Crossref] [PubMed]
- De Ferrari GM, Klersy C, Ferrero P, et al. Atrial fibrillation in heart failure patients: prevalence in daily practice and effect on the severity of symptoms. Data from the ALPHA study registry. Eur J Heart Fail 2007;9:502-9. [Crossref] [PubMed]
- Pilgrim T, Kalesan B, Zanchin T, et al. Impact of atrial fibrillation on clinical outcomes among patients with coronary artery disease undergoing revascularisation with drug-eluting stents. EuroIntervention 2013;8:1061-71. [Crossref] [PubMed]
- Wang TK, Ramanathan T, Choi DH, et al. Preoperative atrial fibrillation predicts mortality and morbidity after aortic valve replacement. Interact Cardiovasc Thorac Surg 2014;19:218-22. [Crossref] [PubMed]
- Mojoli M, Gersh BJ, Barioli A, et al. Impact of atrial fibrillation on outcomes of patients treated by transcatheter aortic valve implantation: A systematic review and meta-analysis. Am Heart J 2017;192:64-75. [Crossref] [PubMed]
- Sannino A, Gargiulo G, Schiattarella GG, et al. A meta-analysis of the impact of pre-existing and new-onset atrial fibrillation on clinical outcomes in patients undergoing transcatheter aortic valve implantation. EuroIntervention 2016;12:e1047-e56. [Crossref] [PubMed]
- HCUP Databases. Healthcare Cost and Utilization Project (HCUP). Agency for Healthcare Research and Quality. Available online: https://www.hcup-us.ahrq.gov/nisoverview.jsp. Published 2018.
- Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol 1992;45:613-9. [Crossref] [PubMed]
- Piccini JP, Hammill BG, Sinner MF, et al. Incidence and prevalence of atrial fibrillation and associated mortality among Medicare beneficiaries, 1993-2007. Circ Cardiovasc Qual Outcomes 2012;5:85-93. [Crossref] [PubMed]
- January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol 2014;64:e1-76. [Crossref] [PubMed]
- Wang J, Yu W, Zhou Y, et al. Independent Risk Factors Contributing to Acute Kidney Injury According to Updated Valve Academic Research Consortium-2 Criteria After Transcatheter Aortic Valve Implantation: A Meta-analysis and Meta-regression of 13 Studies. J Cardiothorac Vasc Anesth 2017;31:816-26. [Crossref] [PubMed]
- Najjar M, Salna M, George I. Acute kidney injury after aortic valve replacement: incidence, risk factors and outcomes. Expert Rev Cardiovasc Ther 2015;13:301-16. [Crossref] [PubMed]
- Arbel Y, Zivkovic N, Mehta D, et al. Factors associated with length of stay following trans-catheter aortic valve replacement - a multicenter study. BMC Cardiovasc Disord 2017;17:137. [Crossref] [PubMed]
- Kamioka N, Wells J, Keegan P, et al. Predictors and Clinical Outcomes of Next-Day Discharge After Minimalist Transfemoral Transcatheter Aortic Valve Replacement. JACC Cardiovasc Interv 2018;11:107-15. [Crossref] [PubMed]
- Stachon P, Kaier K, Zirlik A, et al. Risk factors and outcome of postoperative delirium after transcatheter aortic valve replacement. Clin Res Cardiol 2018;107:756-62. [Crossref] [PubMed]
- Wu HY, Sahadevan S, Ding YY. Factors associated with functional decline of hospitalised older persons following discharge from an acute geriatric unit. Ann Acad Med Singapore 2006;35:17-23. [PubMed]
- Tarantini G, Mojoli M, Windecker S, et al. Prevalence and Impact of Atrial Fibrillation in Patients With Severe Aortic Stenosis Undergoing Transcatheter Aortic Valve Replacement: An Analysis From the SOURCE XT Prospective Multicenter Registry. JACC Cardiovasc Interv 2016;9:937-46. [Crossref] [PubMed]
- Abubakar H, Yassin AS, Akintoye E, et al. Financial Implications and Impact of Pre-existing Atrial Fibrillation on In-Hospital Outcomes in Patients Who Underwent Transcatheter Aortic Valve Implantation (from the National Inpatient Database). Am J Cardiol 2018;121:1587-92. [Crossref] [PubMed]
- Urena M, Hayek S, Cheema AN, et al. Arrhythmia burden in elderly patients with severe aortic stenosis as determined by continuous electrocardiographic recording: toward a better understanding of arrhythmic events after transcatheter aortic valve replacement. Circulation 2015;131:469-77. [Crossref] [PubMed]


