Overweight and obesity: an emerging problem in patients with congenital heart disease
Introduction
Congenital heart defects (CHD) are among the most common types of birth defects. Worldwide, CHD affects 1.35–1.5 million children each year. More than 90% of them reach adulthood due to the developments and successes of modern medicine (1). Nevertheless, almost all of these patients are chronically ill and suffer from residua and sequelae of the underlying cardiac disorder. Empirical evidence suggests that a substantial number of the affected patients engage in unhealthy lifestyle behaviors, such as smoking, physical inactivity and bad nutritional habits (2). Furthermore, there is increasing evidence that CHD patients have a higher susceptibility to cardiac and non-cardiac comorbidities, such as obesity and metabolic disorders (2-4). This review elaborates upon the challenging constraints and contradictions which increasingly accompany CHD patients and complicate their course of disease.
Overweight and obesity are among the most significant contributors to illness and adverse health outcomes including metabolic syndrome, diabetes mellitus and cardiovascular disease (5,6). Causes for excess weight in adulthood can be traced back to early childhood as a high childhood BMI is a predictor for overweight and obesity later in life (7). Over the past decades, a considerable increase in the prevalence of childhood obesity has been documented worldwide. According to recent statistics released by the WHO, the global number of overweight or obese children amounted from 32 million in 1990 to 41 million in 2016 and current trends continue to rise (8). This epidemic of obesity is proliferating among the population and children with CHD are exhibiting its adverse health effects.
Studies confirm that more than a quarter of CHD patients are overweight (9,10). The causes of this situation are multi-faceted and complex and the current treatment strategies are fractured. Restricted physical activity and excessive feeding in early infancy have been identified as major causes for excess weight in this population (11). In fact, the problem starts with the increased risk of mal- and undernutrition in CHD children due to increased metabolic rates, malabsorption and other additive effects, such as hypoxia and pulmonary hypertension (12). Consequently, most treatment strategies prioritize adequate growth and weight development by prescribing increased caloric intake regardless of the diverse range of cardiac lesion types and varying individual medical needs (13,14). Although nutritional needs and physical functional capacity change after successful surgery and throughout the lifespan, inappropriate dietary behaviors and physical inactivity often persist across childhood and adolescence among patients with CHD (15). The family system frequently exacerbates unhealthy behaviors both directly, by excessive feeding and restricting sports activities, and indirectly, by setting unhealthy role models (16-18). Individual eating behavior can be conceptualized as a lifelong learning process which is shaped by our social and cultural environment as well as personal ingrained habits (19). From this perspective, obesity among CHD patients constitutes a multi-factorial problem which involves traditional risk factors as well as factors inherent to CHD including overprotective parenting (OP), poor role modelling and increased psychological distress in this given population (15,20).
The present review focuses on the myriad psychosocial effects resulting from the diagnosis of CHD and their impact on the risk of developing obesity thus exacerbating the existing implications and challenges of CHD patients. It seeks to identify the numerous unique risk factors of developing obesity in CHD patients and provides relevant explanations associated with the increasing prevalence of obesity among CHD patients. As obesity and overweight pose an additional cardiovascular burden to patients with CHD, a thorough understanding of the risks for obesity in CHD children may have a significant long-term impact on their care and preventative measures. Holistic primary prevention of excess weight is of utmost importance in the growing population of CHD patients. It is suggested that healthcare providers need to address weight problems in CHD patients in a comprehensive way by approaching the issue from diverse angles while still keeping in mind the nutritional challenges CHD patients are facing in their recovery.
Epidemiology of obesity in CHD patients
The topic of obesity among pediatric and adult CHD populations has only recently emerged and prevalence rates have rarely been reported. Figure 1 provides prevalence rates of obesity and overweight in CHD patients.
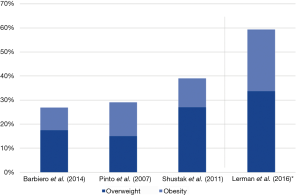
The first cross-sectional study to recognize the epidemic of obesity among CHD children was conducted by Pinto et al. in a population of 1,523 children with various forms of CHD aged 6–19 years from Boston and Philadelphia (21). Obesity (BMI ≥95th percentile) and overweight (BMI ≥85–95th percentile) were classified according to the CDC (US Centers for Disease Control and Prevention) guidelines (22). The population reflected a large spectrum of heart defects and was divided into subgroups according to their severity. The combined prevalence of obesity and overweight among the cohort was 29% and did not differ from national data when compared with the concurrent National Health and Nutrition Examination Survey (NHANES). Patients with complex CHD had the lowest prevalence of overweight and obesity (15.9%) which can be explained by residua, sequelae and ongoing complications from the underlying CHD (3,23). Secondary effects, such as increased systolic blood pressure in obese and overweight patients with CHD, were also documented in this study (21).
Another retrospective cross-sectional study estimated the overall prevalence of overweight and obesity at 30% among a New York cohort of pediatric CHD patients (24). Again, prevalence rates for overweight and obesity were similar to the healthy population rates for overweight and obesity. The authors concluded that CHD children are similarly vulnerable to the global obesity epidemic as the general population. But they found significant differences based on gender and ethnicity with Hispanic ethnicity and male gender presenting the highest risk for the development of obesity. Furthermore, surgical status was found to have an impact on weight as surgical males were twice as likely to be obese than their non-surgical counterparts (risk of obesity: 24% in postsurgical males vs. 11% in non-surgical counterparts, OR: 2.2). This subgroup of patients may particularly benefit from lifestyle interventions, such as dietary counseling and physical activity, as a routine part of their follow-up care. In line with Pinto et al. (21) the prevalence of obesity among complex CHD patients was found to be low compared to simple defects (6% in complex univentricular heart disease vs. 18% in simple defects) (24).
A more recent study confirmed that 26.9% of adolescents with CHD are affected by excess weight (defined as a BMI ≥85th percentile) (9). Additionally, the authors observed modifiable risk factors for ischemic heart disease among their CHD patient population, including altered lipid profiles (high cholesterol/LDL/triglycerides), sedentary lifestyle and a positive family history of obesity. Since children with CHD are prone to heart disease issues to begin with, addressing diet-related risk factors is particularly important in this patient population.
Currently, the number of adults with CHD (ACHD) exceeds the number of children with CHD and is expected to grow by 5% per year (1). According to Pearson et al., obesity is a great concern among ACHD (>18 years of age), as 54% of them have a BMI >25 kg/m2 (25). A recent large-scale study by Lerman et al. confirmed that 59.5% out of 1,451 ACHD were overweight to morbidly obese (26). Their cohort was divided into three age groups, younger (19–44 years), middle aged (45–64 years), and senior aged patients (>65 years). The frequencies of obesity among ACHD did not differ across all age divisions when compared with their matched controls. Although there is little evidence on how obesity may affect ACHD, it is reasonable to believe that obesity will adversely influence health outcomes (i.e., heart failure, pulmonary hypertension, arrhythmia etc.) in this population.
Etiology of obesity in CHD patients
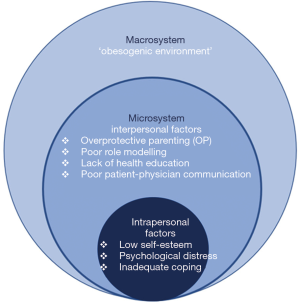
Obesity in CHD patients is the result of an interplay among a complex set of factors which can be divided into micro-level and macro-level factors (Figure 2). Micro-level contributions are defined as the most proximal influences on a child with CHD, like parenting practices, nutritional education and psychological factors (27). Macro-level factors include adverse influences by the “obesogenic environment” (28) due to urbanization and industrialization. Both components are rooted in childhood and affect food choices and lifestyle behaviors in CHD patients throughout life.
Micro-level factors
OP
Parents of children with CHD suffer from stress up to the level of traumatization after the diagnosis of CHD (29). Feelings of sadness, depression, or being overwhelmed can be a normal part of their coping process with the situation and can even lead to “posttraumatic growth” (30). Yet, parental difficulties in coping can potentially impact caregiving processes (29).
Though parenting of children with CHD may be based on the desire for a positive, healthy outcome for their children, the parents’ OP behavior can contribute to additional CHD complications. The challenge for parents is to protect their children adequately but not overprotect them. Research on the impact of childhood chronic illness indicates that mothers tend to adapt overprotective or highly controlling behaviors when it comes to caring for a child with CHD (31). Their OP might be driven by bad conscience, the urge to protect their vulnerable child or as a means of giving love (32). There is compelling evidence that maternal protectiveness is associated with higher odds for child overweight and obesity (33). In the case of CHD children, overprotective parents are more likely to restrict physical exercise (16). At the same time, they may feel pressured to overfeed their children in order to facilitate normal weight and growth development (13). In fact, many mothers view their child’s excess weight as an indicator for health and safety (34). Thus, feeding becomes more than just meeting the child’s physiological needs: food equals love, health and being cared for. Overfeeding in childhood has been shown to be associated with undesirable nutritional outcomes putting children at higher risk for obesity later in life (35).
Parental modelling
Another area which requires additional scrutiny in obesity prevention in CHD children is based on Social Learning Theory (SLT). SLT suggests that our behavior is learned through observing and mimicking others (36). SLT plays a key role in understanding eating habits as they emerge through observational learning and modelling. It is well documented that parental attitudes certainly impact a child’s eating behavior and eating related attitudes (37). As mothers presumably function as nutritional gatekeepers regulating the majority of food available at home (38), their health and eating behaviors have a direct impact on their children’s eating habits and preferences through exposure to certain foods and role modelling (39). Hence, caregivers can influence unhealthy behaviors by setting unhealthy standards themselves. Research indicates that maternal overweight and obesity is not only linked to a child’s excess weight, but also associated with a significantly higher risk for bearing children with CHD (17,18). It can be assumed, that CHD children are more likely to have morbidly overweight mothers and their eating habits are, therefore, tied to these role models. In terms of obesity prevention in CHD patients, it is, therefore, crucial to address the parent’s diet itself as it is the most important source of information and influence on their offspring.
Health education
Proper knowledge on nutrition and physical activity is an essential part of taking responsibility for one’s own health. Research implies that only a minority of CHD patients and their parents are educated on appropriate diet, nutrition and exercising (40-42). According to Lentzner et al. only 59% of cardiologists provide patients with counselling on weight issues, while nutrition and diet were discussed by 63% (41). In another study, mothers of children with CHD demonstrated significant shortcomings in their understanding of the disease and its implications regarding diet and activity restrictions (42). Distorted maternal perceptions of CHD, such as “the bigger the infant, the healthier”, may lead to inappropriate restrictions and child feeding practices which can set the stage for the development of overweight or obesity later in life (43). With specific reference to CHD children, excessive bottle feeding in order to increase caloric intake may impact the newborn’s ability to perceive normal hunger and satiety cues leading to disturbed eating attitudes in the long run (44).
In fact, compared to healthy children, those with CHD are more likely to be overweight due to physical inactivity (21). Nonetheless, non-educated parents tend to promote a sedentary lifestyle for their children as a result of overprotection and the uncertainty as to which extent and intensity physical exercise is recommended for their children (45). In order to lower the risk of future obesity and ischemic heart disease, research indicates a compelling need for healthcare providers to consistently educate and promote healthy lifestyle behaviors in CHD patients in order to reduce modifiable cardiovascular risk factors. Time constraints, the perceived role as a cardiologist and the lack of training in addressing lifestyle recommendations have been identified as the most common barriers to health education in this patient population (46). It is, therefore, essential to establish curricular standards in the field of nutrition and physical activity in order to improve patient-physician communication and education.
Physicians need to provide a concise exercise regimen that fits each patient’s individual purpose in order to encourage a physically active lifestyle. Accordingly, Budt et al. suggest a personalized exercise prescription tailored for each CHD patient via a specific algorithm. It includes the patient’s medical history, a physical examination, an assessment of five baseline parameters (ventricular function, pulmonary artery pressure, aorta, arrhythmia, saturation at rest/during exercise), the type and relative intensity of each exercise as well as cardiopulmonary testing and follow-up. However, due to the heterogeneity of lesions not all patients fit the suggested algorithm and future clinical research is required to maximize the regimen’s health benefits and minimize its cardiovascular risk for CHD patients (45).
Psychological factors
Children with CHD are faced with unique and often complex medical and psychological challenges early in life (47). A substantial number of CHD patients experience significant psychological distress, such as anxiety and depression (47-49). Additionally, patients frequently lack self-esteem and fail to develop autonomous identities as a long-term consequence of OP (50). At the same time, OP is reversely associated with higher levels of children's anxiety and difficulties in coping with stressful social situations. Depression, anxiety and low self-esteem have been identified as predictors for an increased risk of obesity (51,52).
Numerous research findings show that exposure to chronic stress is associated with overweight and obesity (53). Research has identified two possible pathways linking stress and obesity (54). These factors are presumably applicable to the population of CHD patients. First, chronic stress arising from complex psychosocial challenges in CHD patients may alter food intake by promoting the consumption of energy-dense foods. Second, stress-induced sympathetic and neuroendocrine activation contribute to increased fat storage within visceral depots, which in turn amplifies the risk of cardiovascular risk factors (dyslipidemia, type 2 diabetes mellitus, metabolic syndrome) (54). To date, the psychological situation of ACHD and its relation to their health behavior have received little attention. Therefore, screening for early detection and management of psychological issues may empower patients to make wiser decisions in their healthcare and improve long-term outcomes.
Macro-level factors
Obesogenic environment
Our eating patterns are strongly influenced by environmental characteristics. The term ‘obesogenic environment’ refers to the role of environmental factors in determining nutrition and physical activity (55). Key factors such as food processing, pricing and distribution have led to an increased consumption of energy-dense foods. Similarly, changes in the built environment and the technology have decreased physical activity and energy expenditure. These patterns triggered shifts in energy imbalance and led to a global obesity epidemic (56). Unfortunately, the exact same risk factors have had a significant impact on the population of CHD patients as well. A study assessing risk factors for atherosclerosis in pediatric patients with CHD found that many of them adopted unhealthy diets with low fruit and vegetable intake but high consumption of sugar sweetened beverages and high-fat foods. In addition, the cohort spent a large amount of their time sedentary (57). Other findings provide support, that even healthy CHD patients after successful surgery showed significantly less vigorous physical activity levels than their controls (10,58). Consequently, physical inactivity has been shown to be the strongest predictor for excess weight at follow-up (58). As CHD patients already present a high-risk constellation for cardiovascular morbidity and premature mortality, preventive cardiac care to protect them from the dangers of obesity should be given a prominent role. The NCEP (National Cholesterol Education Program) recommends a dual strategy that combines a population approach and an individualized approach to lower blood cholesterol levels in CHD patients (59).
The importance of nutrition for the CHD population: the gut microbiome
The important role that gut microbes contribute in the development of cardiovascular disease has recently received attention (60-62). Gut microbes represent a filter of our greatest environmental exposure which is our nutrition (61). Accordingly, dietary intake leads to significant changes in the composition of gut microbiota. Previous research has particularly focused on trimethylamine N-oxide (TMAO) which is generated from choline, lecithine and L-carnitine—compounds found in dairy products, eggs, fish and red meat—by gut microbial metabolism (61). Apparently, there is a positive correlation between elevated levels of TMAO and an increased risk for adverse cardiovascular events and premature death (60). This correlation highlights the role of diet and nutrition in the prevention of CHD.
The TMAO pathway is only one out of many diet-related microbial pathways that can potentially be linked to adverse cardiovascular outcomes (61). However, research in this area is still insufficient. Preliminary findings in this field reveal new, important insights of how nutrition is linked to cardiovascular health. Further research is needed to identify the distinct microbiotic composition and its implications in terms of dietary prevention in the population of CHD patients. In this regard, plant-based diets may become increasingly recognized due to demonstrated lower levels of TMAO in vegetarian/vegan participants compared to omnivorous subjects (63).
Conclusions
Due to medical and technological advances, an increasing number of children with CHD survives into adulthood. In addition to common risk factors for overweight and obesity, CHD patients often face unique psychosocial challenges (OP, psychological distress, lack of health education) which may put them at higher risk for developing disordered eating patterns and excess weight. Unhealthy lifestyle habits and obesity contribute to the development of additional health burdens which may adversely impact the per-se fragile cardiovascular condition.
This paper is the first attempt to put diet, nutrition and CHD into context and seek explanations for problematic health behaviors in this population. The findings not only provide a clearer picture of problematic influences on health behaviors among CHD patients, but also help to seize possible opportunities in preventive cardiac care. Preventive cardiac care should be emphasized in order to empower patients to make wise decisions in their healthcare particularly when it comes to healthy nutrition and exercise. Given the alarming rates of excess weight among CHD patients, assessments of diet and physical activity should become an integral part of routine care. Since children with CHD solely rely on parental guidance, parents need to be adequately educated and trained in taking care for their cardiopathic child in order to prevent common weight misperceptions. In addition to nutritional guidance, psychological counselling might be necessary for both, parents and patients, to reduce psychological distress and facilitate coping with the disease.
Acknowledgments
Funding: This work was supported by an unrestricted research grant from the patient organization “Herzkind e. V.” and the German Heart Foundation (“Deutsche Herzstiftung e.V.”) [grant number F-30-15].
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Stout KK, Daniels CJ, Aboulhosn JA, et al. 2018 AHA/ACC guideline for the management of adults with congenital heart disease: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Circulation 2018. Available online: https://doi.org/ [Crossref]
- Moons P, Deyk KV, Dedroog D, et al. Prevalence of cardiovascular risk factors in adults with congenital heart disease. Eur J Cardiovasc Prev Rehabil 2006;13:612-6. [Crossref] [PubMed]
- Neidenbach RC, Lummert E, Vigl M, et al. Non-cardiac comorbidities in adults with inherited and congenital heart disease: report from a single center experience of more than 800 consecutive patients. Cardiovasc Diagn Ther 2018;8:423-31. [Crossref] [PubMed]
- Deen JF, Krieger EV, Slee AE, et al. Metabolic syndrome in adults with congenital heart disease. J Am Heart Assoc 2016;5:e001132. [Crossref] [PubMed]
- Kopelman P. Health risks associated with overweight and obesity. Obes Rev 2007;8:13-7. [Crossref] [PubMed]
- Franks PW, Hanson RL, Knowler WC, et al. Childhood obesity, other cardiovascular risk factors, and premature death. N Engl J Med 2010;362:485-93. [Crossref] [PubMed]
- Evensen E, Wilsgaard T, Furberg AS, et al. Tracking of overweight and obesity from early childhood to adolescence in a population-based cohort–the Tromsø Study, Fit Futures. BMC Pediatr 2016;16:64. [Crossref] [PubMed]
- WHO. Report of the Commission on Ending Childhood Obesity: implementation plan: executive summary. 2017.
- Barbiero SM, Sica CDA, Schuh DS, et al. Overweight and obesity in children with congenital heart disease: combination of risks for the future? BMC Pediatr 2014;14:271. [Crossref] [PubMed]
- Cohen MS. Clinical practice: the effect of obesity in children with congenital heart disease. Eur J Pediatr 2012;171:1145-50. [Crossref] [PubMed]
- Müller-Riemenschneider F, Nocon M, Willich SN. Prevalence of modifiable cardiovascular risk factors in German adolescents. Eur J Cardiovasc Prev Rehabil 2010;17:204-10. [Crossref] [PubMed]
- Varan B, Tokel K, Yilmaz G. Malnutrition and growth failure in cyanotic and acyanotic congenital heart disease with and without pulmonary hypertension. Arch Dis Child 1999;81:49-52. [Crossref] [PubMed]
- Forchielli ML, McColl R, Walker WA, et al. Children with congenital heart disease: a nutrition challenge. Nutr Rev 1994;52:348-53. [Crossref] [PubMed]
- Nydegger A, Bines JE. Energy metabolism in infants with congenital heart disease. Nutrition 2006;22:697-704. [Crossref] [PubMed]
- Moola F, Fusco C, Kirsh JA. The perceptions of caregivers toward physical activity and health in youth with congenital heart disease. Qual Health Res 2011;21:278-91. [Crossref] [PubMed]
- Reybrouck T, Mertens L. Physical performance and physical activity in grown-up congenital heart disease. Eur J Cardiovasc Prev Rehabil 2005;12:498-502. [Crossref] [PubMed]
- Brite J, Laughon SK, Troendle J, et al. Maternal overweight and obesity and risk of congenital heart defects in offspring. Int J Obes (Lond) 2014;38:878-82. [Crossref] [PubMed]
- Mills JL, Troendle J, Conley MR, et al. Maternal obesity and congenital heart defects: a population-based study. Am J Clin Nutr 2010;91:1543-9. [Crossref] [PubMed]
- Ellrott T. Psychologische Aspekte der Ernährung. Diabetologie und Stoffwechsel 2013;8:R57-R70. [Crossref]
- Parsons TJ, Power C, Logan S, et al. Childhood predictors of adult obesity: a systematic review. Int J Obes Relat Metab Disord 1999;23 Suppl 8:S1-107. [PubMed]
- Pinto NM, Marino BS, Wernovsky G, et al. Obesity is a common comorbidity in children with congenital and acquired heart disease. Pediatrics 2007;120:e1157-e1164. [Crossref] [PubMed]
- Hedley AA, Ogden CL, Johnson CL, et al. Prevalence of overweight and obesity among US children, adolescents, and adults, 1999-2002. JAMA 2004;291:2847-50. [Crossref] [PubMed]
- Singh S, Desai R, Fong HK, et al. Extra-cardiac comorbidities or complications in adults with congenital heart disease: a nationwide inpatient experience in the United States. Cardiovasc Diagn Ther 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Shustak RJ, McGuire SB, October TW, et al. Prevalence of obesity among patients with congenital and acquired heart disease. Pediatr Cardiol 2012;33:8-14. [Crossref] [PubMed]
- Pearson D, Rodriguez E, Fernandez S. Prevalence of obesity in adults with congenital heart disease [abstract]. Presented at Fifth National Conference Adult Congenital Heart Association. Philadelphia, PA, 2008.
- Lerman JB, Parness IA, Shenoy RU. Body Weights in Adults With Congenital Heart Disease and the Obesity Frequency. Am J Cardiol 2017;119:638-42. [Crossref] [PubMed]
- Rosenkranz RR, Dzewaltowski DA. Model of the home food environment pertaining to childhood obesity. Nutr Rev 2008;66:123-40. [Crossref] [PubMed]
- Egger G, Swinburn B. An "ecological" approach to the obesity pandemic. BMJ: British Medical Journal 1997;315:477. [Crossref] [PubMed]
- Farley LM, DeMaso DR, D'Angelo E, et al. Parenting stress and parental post-traumatic stress disorder in families after pediatric heart transplantation. J Heart Lung Transplant 2007;26:120-6. [Crossref] [PubMed]
- Soulvie MA, Desai PP, White CP, et al. Psychological distress experienced by parents of young children with congenital heart defects: A comprehensive review of literature. J Soc Serv Res 2012;38:484-502. [Crossref]
- Power TG, Dahlquist LM, Thompson SM, et al. Interactions between children with juvenile rheumatoid arthritis and their mothers. J Pediatr Psychol 2003;28:213-21. [Crossref] [PubMed]
- Mullins LL, Wolfe-Christensen C, Hoff Pai AL, et al. The relationship of parental overprotection, perceived child vulnerability, and parenting stress to uncertainty in youth with chronic illness. J Pediatr Psychol 2007;32:973-82. [Crossref] [PubMed]
- Hancock KJ, Lawrence D, Zubrick SR. Higher maternal protectiveness is associated with higher odds of child overweight and obesity: a longitudinal Australian study. PLoS One 2014;9:e100686. [Crossref] [PubMed]
- Kaufman L, Karpati A. Understanding the sociocultural roots of childhood obesity: food practices among Latino families of Bushwick, Brooklyn. Soc Sci Med 2007;64:2177-88. [Crossref] [PubMed]
- Patrick H, Nicklas TA, Hughes SO, et al. The benefits of authoritative feeding style: caregiver feeding styles and children's food consumption patterns. Appetite 2005;44:243-9. [Crossref] [PubMed]
- Bandura A. Self-referent mechanisms in social learning theory. American Psychologist 1979;34:439-41. [Crossref]
- Brown R, Ogden J. Children’s eating attitudes and behaviour: a study of the modelling and control theories of parental influence. Health Educ Res 2004;19:261-71. [Crossref] [PubMed]
- Wansink B. Nutritional gatekeepers and the 72% solution. J Am Diet Assoc 2006;106:1324-7. [Crossref] [PubMed]
- Oliveria SA, Ellison RC, Moore LL, et al. Parent-child relationships in nutrient intake: the Framingham Children's Study. Am J Clin Nutr 1992;56:593-8. [Crossref] [PubMed]
- Neidenbach R, Kaemmerer H, Pieper L, et al. Striking supply gap in adults with congenital heart disease? Dtsch Med Wochenschr 2017;142:301-3. [PubMed]
- Lentzner BJ, Connolly DM, Phoon CK. Do paediatric cardiologists discuss cardiovascular risk factors with patients and their families? Cardiol Young 2003;13:551-8. [Crossref] [PubMed]
- Kaden GG, McCarter RJ, Johnson SF, et al. Physician-patient communication: understanding congenital heart disease. Am J Dis Child 1985;139:995-9. [Crossref] [PubMed]
- Baughcum AE, Burklow KA, Deeks CM, et al. Maternal feeding practices and childhood obesity: a focus group study of low-income mothers. Arch Pediatr Adolesc Med 1998;152:1010-4. [Crossref] [PubMed]
- Birch LL, Zimmerman SI, Hind H. The influence of social-affective context on the formation of children's food preferences. Journal of Nutrition Education 1981;13:S115-S118. [Crossref]
- Budts W, Börjesson M, Chessa M, et al. Physical activity in adolescents and adults with congenital heart defects: individualized exercise prescription. Eur Heart J 2013;34:3669-74. [Crossref] [PubMed]
- Kris-Etherton PM, Akabas SR, Bales CW, et al. The need to advance nutrition education in the training of health care professionals and recommended research to evaluate implementation and effectiveness. Am J Clin Nutr 2014;99:1153S-66S. [Crossref] [PubMed]
- Andonian C. Current research status on the psychological situation of adults with congenital heart disease. Cardiovasc Diagn Ther 2018;8:799-804. [Crossref] [PubMed]
- Pauliks LB. Depression in adults with congenital heart disease-public health challenge in a rapidly expanding new patient population. World J Cardiol 2013;5:186. [Crossref] [PubMed]
- Jackson JL, Leslie CE, Hondorp SN. Depressive and anxiety symptoms in adult congenital heart disease: Prevalence, health impact and treatment. Prog Cardiovasc Dis 2018;61:294-9. [Crossref] [PubMed]
- Cohen M, Mansoor D, Langut H, et al. Quality of life, depressed mood, and self-esteem in adolescents with heart disease. Psychosom Med 2007;69:313-8. [Crossref] [PubMed]
- Bradley RH, Houts R, Nader PR, et al. The relationship between body mass index and behavior in children. J Pediatr 2008;153:629-34. [Crossref] [PubMed]
- Chung KH, Chiou HY, Chen YH. Psychological and physiological correlates of childhood obesity in Taiwan. Sci Rep 2015;5:17439. [Crossref] [PubMed]
- Scott KA, Melhorn SJ, Sakai RR. Effects of chronic social stress on obesity. Curr Obes Rep 2012;1:16-25. [Crossref] [PubMed]
- Razzoli M, Bartolomucci A. The dichotomous effect of chronic stress on obesity. Trends Endocrinol Metab 2016;27:504-15. [Crossref] [PubMed]
- Townshend T, Lake A. Obesogenic environments: current evidence of the built and food environments. Perspect Public Health 2017;137:38-44. [Crossref] [PubMed]
- Popkin BM, Duffey K, Gordon-Larsen P. Environmental influences on food choice, physical activity and energy balance. Physiol Behav 2005;86:603-13. [Crossref] [PubMed]
- Massin MM, Hövels-Gürich H, Seghaye MC. Atherosclerosis lifestyle risk factors in children with congenital heart disease. Eur J Cardiovasc Prev Rehabil 2007;14:349-51. [Crossref] [PubMed]
- Stefan MA, Hopman WM, Smythe JF. Effect of activity restriction owing to heart disease on obesity. Arch Pediatr Adolesc Med 2005;159:477-81. [Crossref] [PubMed]
- National Cholesterol Education Program. report of the expert panel on blood cholesterol levels in children and adolescents. Pediatrics 1992;89:525-84. [PubMed]
- Li XS, Obeid S, Klingenberg R, et al. Gut microbiota-dependent trimethylamine N-oxide in acute coronary syndromes: a prognostic marker for incident cardiovascular events beyond traditional risk factors. Eur Heart J 2017;38:814-24. [PubMed]
- Tang WH, Hazen SL. The gut microbiome and its role in cardiovascular diseases. Circulation 2017;135:1008-10. [Crossref] [PubMed]
- Velasquez MT, Ramezani A, Manal A, et al. Trimethylamine N-oxide: the good, the bad and the unknown. Toxins 2016;8:326. [Crossref] [PubMed]
- Koeth RA, Wang Z, Levison BS, et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 2013;19:576. [Crossref] [PubMed]