Four-year clinical follow-up of the first-in-man randomized comparison of a novel sirolimus eluting stent with abluminal biodegradable polymer and ultra-thin strut cobalt-chromium alloy: the INSPIRON-I trial
Introduction
Earlier-generation drug-eluting stents (DES) were highly effective in reducing in-stent neointimal formation and therefore the occurrence of restenosis (1). However, there are some concerns about a related thrombogenic response, leading to late and very late stent thrombosis (ST) (2). Permanent polymers have been implicated in suboptimal polymer biocompatibility, persistent inflammatory reaction, and delayed healing observed after DES (3,4). In addition, thick-strut stents, such as first-generation DES, have been demonstrated to be more thrombogenic than identical thin-strut comparators in ex vivo and experimental models (5).
Newer DES stent technologies have been developed to resolve these issues, such as the use of stents with thinner struts and the use of biodegradable coatings to improve biocompatibility. These properties might confer an additional benefit in terms of re-endothelialisation and restenosis (6,7), especially when the coating is applied only at the abluminal surface of the stent, leaving the luminal side of the struts free of drug and polymeric coating.
The Inspiron™ (Scitech Medical™, Aparecida de Goiânia, Goiás, Brazil) sirolimus-eluting stent (SES) uses an ultra-thin L-605 cobalt-chromium alloy with a 75 µm strut thickness platform coated with a matrix of a biodegradable polymer (BP) and sirolimus applied to the abluminal surface, causing a significantly lower concentration of drug in the vessel (8). The polymer is degraded by hydrolysis and enzymatic action and then eliminated from the body through the tricarboxylic acid cycle in 6-9 months, releasing 80% of the drug (sirolimus) within 30 days after its deployment.
The initial results of the first-in-man (8) comparison of the novel Inspiron™ SES and its equivalent Cronus™ bare metal stent (BMS) showed its effectiveness in reducing restenosis at 6 months (8). Despite the small sample size and the consequent lack of power to support definitive statements, the present study aims at evaluating the long-term clinical outcomes of patients treated with this novel SES enrolled into the INSPIRON-I trial.
Methods
Study design and population—the INSPIRON-I trial
Patients were enrolled in four centres in Brazil, from January 2009 to December 2010, and randomized in a 2:1 basis for percutaneous coronary intervention (PCI) using the novel Inspiron™ DES or the Cronus™ BMS. Balloon angioplasty and stent implantation were performed according to standard techniques.
Patients were eligible for enrollment if they were aged 18 years or above, had symptoms of angina pectoris and/or documented (inducible or spontaneous) ischaemia. The angiographic inclusion criteria were non-occlusive de novo coronary lesions <20 mm in length and reference diameter of 2.5-3.5 mm. The exclusion criteria were cardiogenic shock, left ventricular ejection fraction <30%, lesions requiring two or more stents, ostial lesions, major surgery within 6 weeks before planned stenting procedure, stenosis ≥50% of the left main coronary artery, angiographic evidence of thrombus or poor distal flow at the lesion site, lesion at a junction of a side branch with a diameter ≥2 mm, staged procedure planned within 3 months of the index procedure, known allergy to the study medications including sirolimus, and pregnancy (present, suspected, or planned) (1).
The study complied with the Declaration of Helsinki and was approved by all institutional ethics committees. All patients provided written informed consent for participation in the trial.
Study stents
The Cronus™ BMS and Inspiron™ SES (both by Scitech Medical™, Aparecida de Goiânia, Goiás, Brazil) use an ultra-thin L-605 cobalt-chromium alloy, with 75 µm strut thickness. The stent was designed in corrugated rings, with a maximum angle between the rods of 45° in its expanded form. These rings are made of short cells (1.1 mm), which allows for increasing the number of cells then improving the homogeneity of metal cover in angulated and curved segments. The connection between rings is performed using short-shaped waveform links (65 µm) which provides a circumferential maximum diameter area of approximately 6 mm, in order to facilitate access to lateral branches (Figure 1).
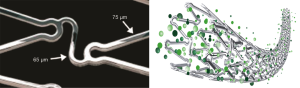
The Inspiron™ SES has a novel platform with an abluminal BP, which is a blend of poly L-lactic acid (PLLA) and poly DL-lactic-co-glycolic acid (PDLLGA). It is degraded by hydrolysis and enzymatic action to monomeric acids and eliminated from the body through the tricarboxylic acid cycle that releases 80% of the drug within 30 days (data from Scitech Medical™, Aparecida de Goiânia, Goiás, Brazil).
Clinical follow-up
Baseline and procedural characteristics were recorded in a dedicated database for all patients. Angiographic and intravascular ultrasound (IVUS) follow-up were performed at 6 months and have been previously reported (8). Adverse events were assessed in the hospital and clinical follow-up was performed at 1, 6, 9, 12, 24, 36 and 48 months, as initially planned.
Study endpoints and definitions
Late clinical follow-up evaluated the occurrence of major adverse cardiac events (MACE), defined as the composite of death, myocardial infarction (MI) and/or clinically driven surgical or percutaneous revascularization of the target lesion or vessel up to 48 months after the index procedure. ST was defined according to the Academic Research Consortium definition (9).
Statistical analyses
All data were analysed on the per-treatment evaluable population, and according to the received treatment. Discrete variables are reported as frequencies (%), and continuous variables as mean ± SD. Student’s t-tests were used for continuous variables and Chi-squared or Fisher’s exact tests for categorical variables. For all analyses, a two-sided P=0.05 was considered statistically significant. All analyses were conducted using the statistical package R version 2.13.0 (R Core Team, Vienna, Austria) and the SPSS 20 (IBM Corp, Armonk, NY, USA). Kaplan-Meier curves were used to estimate the incidence of clinical adverse events.
Results
Baseline characteristics
A total of 59 patients were randomized. One patient did not receive the study stents because the target vessel was shown to be totally occluded at the index procedure (due to disease progression between the diagnostic angiogram and the study procedure). Another one was excluded from final analysis because of inadvertent implantation of a non-study stent at the target segment. Therefore, 57 patients (38 in the Inspiron and 19 in the Cronus group) and 60 lesions (39 and 21, respectively) were included for final endpoint analysis.
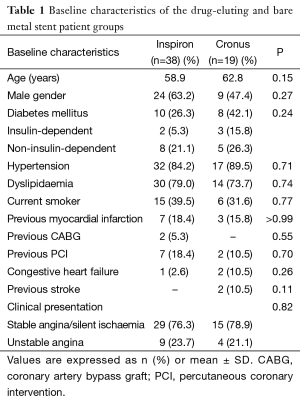
Patients’ characteristics did not show any statistically significant difference between the two groups (Table 1). Overall, the mean age was 60.2±9.5 years and the majority was men (57.9%). Diabetes was present in 31.6%. Most patients had stable coronary disease (stable angina in 63.2% and silent ischemia in 14.0%).
Full table
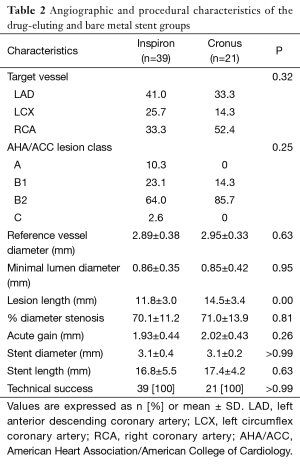
Procedural characteristics, presented in Table 2, were also similar between both groups. Right coronary artery PCI was performed in 40.0% of the patients, left anterior descending in 38.3%, and left circumflex in 21.7%. The mean vessel diameter was 2.9±0.37 mm and the mean minimal luminal diameter was 0.86±0.37 mm. The only difference between groups was the lesion length, shorter in the SES group (11.8±3.0 vs. 14.5±3.4, P=0.004). However, comparing the stent diameter and length between SES and BMS, they were similar in both groups.
Full table
Long term major adverse cardiac events (MACE)
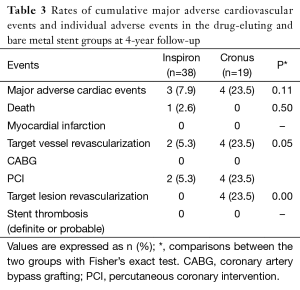
Up to 48 months, clinical follow-up was available in 53 patients (93%). Cumulative event rates were similar between groups, as showed in the Table 3.
Full table
The cumulative incidence of MACE [death, MI, and target vessel revascularization (TVR)] was 7.9% in the Inspiron group vs. 23.5% in the Cronus group (P=0.2). There was one death in the Inspiron arm (2 years after the index procedure) due to an acute pulmonary edema, without documentation of acute MI. There were two TVRs in the Inspiron group but neither of them was a target lesion revascularization (TLR). Conversely, all of the four TVRs in the Cronus group were in the segment of the index procedure and, therefore, TLRs. There was a strong trend toward lower TVR (5.3% vs. 23.5%, P=0.05) and significant lower TLR (0% vs. 23.5%, P=0.02) favoring the Inspiron. No acute MI or (definite/probable) ST occurred in either group at the 48-month follow-up.
The Kaplan-Meier curves of MACE and TLR are plotted in the Figure 2.

Discussion
The main findings of the present study are that up to 4-year clinical follow-up the incidence of MACE was not different between the groups SES and BMS, with significant lower TVR and TLR for Inspiron and no definite/probable ST.
The INSPIRON-I trial (8) tested a novel SES (Inspiron™) which combines a thin cobalt-chromium alloy platform, an abluminal BP and a known efficacious drug to inhibit neointimal hyperplasia, in comparison to its analogue BMS (Cronus™). Initial findings showed that at 6 months the in-segment late lumen loss (LLL) and the percent neointimal obstruction were significantly reduced in the Inspiron group. The SES arm also presented lower in-stent LLL, diameter of stenosis, and binary restenosis, as well as greater minimal luminal diameter. IVUS data at 6 months showed significantly lower hyperplasia volume and less percent neointimal obstruction favoring the Inspiron group (8).
The concerns about the safety of first-generation DES lead to the development of newer devices, with improved metallic platform and polymer technologies. In experimental and clinical scenarios, thin-strut stents have been shown to produce less neointimal hyperplasia than thick-strut BMS (6,7). Thick-strut stents are related to increased thrombogenicity as compared to identical thin-strut counterparts in ex vivo and in vivo models (5). In the Intracoronary Stenting and Angiographic Results: Strut Thickness Effect on Restenosis Outcome (ISAR-STEREO) trials (10,11), thin-strut (50 µm) stents elicited less restenosis than thick-strut (140 µm) BMS.
It is also known that local arterial injury due to durable polymers (DP) might be related to late and very late ST. DP-DES thrombogenicity could potentially be increased at regions of stent surface irregularities, due to displacement or inhomogeneous distribution of the polymer coating. Coarse DES coating irregularities might promote inflammatory reactions, a direct nidus for platelet activation and ST (12).
BP-coated stents may improve safety outcomes as both polymer and drug disappear after the full anti-restenosis effect has been achieved, then avoiding unwelcome inflammatory side effects. The design of exclusively abluminal coating allows additional lower drug concentration and might lead to less vascular inflammation (3,4).
Our findings are in line with those of recent large studies showing the efficacy and the safety outcomes, especially by low risk of very late ST, after BP-DES implantation.
Pooled individual patient data from three large-scale multicentre randomised clinical trials [ISAR-TEST 3 (13), ISAR-TEST 4 (14), and LEADERS (15)] comparing BP-DES with the leading first-generation DP-SES have demonstrated that the first produced a significantly lower risk of TVR and ST up to 4 years, mainly by decreased risk of very late ST (16).
The same authors analysed the data of 497 ST-segment elevation MI patients from this same pooled database. BP-DES significantly improved clinical outcomes up to 4 years. There was a significant reduction of TLR and a trend towards reduction of cardiac death or MI, with non-significant numerically lower ST (17).
In a recent comprehensive network meta-analysis (data from 89 trials including 85,490 patients), Palmerini et al. (18) concluded that BP-biolimus eluting stents were associated with superior clinical outcomes compared to BMS and first-generation DP-DES, and similar rates of all-cause death, cardiac death, MI and TVR compared to other second-generation DP-DES, but higher rates of definite ST than cobalt-chromium everolimus eluting stent.
The recent randomized PAINT trial was designed to evaluate the safety and efficacy of two BP-DES (sirolimus and paclitaxel), compared to the identical BMS platform (19-21). Similar to our results, up to the late 5-year clinical follow-up both BP-DES sustained their effectiveness in reducing re-intervention (TVR and TLR) without an increase in ST (22).
Despite the fact that INSPIRON-I trial was powered for LLL and not for clinical events, there was a reduction in clinical restenosis driven by significantly lower TVR and mainly TLR sustained up to the late 4-year follow up, with similar low rates of MACE, and no occurrence of early or late ST.
These findings demonstrate the feasibility by the early and late clinical safety and efficacy of this novel SES with abluminal BP and thin strut cobalt-chromium alloy platform.
Conclusions
The novel Inspiron™ SES showed a sustained safe and effective clinical profile after 4-year of follow-up, with very low adverse events and null ST occurrence. The INSPIRON-I trial (8) was designed to minimize the number of patients exposed to this novel device, while still providing adequate data about stent safety and efficacy. There are baseline clinical and angiographic statistically non-significant differences between the groups, but the small number of patients does not allow us to draw firm conclusions. Larger trials comparing this novel SES with other BP-DES are necessary to evaluate long term clinical results.
Acknowledgements
Scitech Medical™ (Aparecida de Goiânia, Goiás, Brazil) provided the study stents and the funds to cover the expenses with the angiographic core laboratory.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Kirtane AJ, Gupta A, Iyengar S, et al. Safety and efficacy of drug-eluting and bare metal stents: comprehensive meta-analysis of randomized trials and observational studies. Circulation 2009;119:3198-206. [PubMed]
- Nakazawa G, Finn AV, Joner M, et al. Delayed arterial healing and increased late stent thrombosis at culprit sites after drug-eluting stent placement for acute myocardial infarction patients: an autopsy study. Circulation 2008;118:1138-45. [PubMed]
- Byrne RA, Joner M, Kastrati A. Polymer coatings and delayed arterial healing following drug-eluting stent implantation. Minerva Cardioangiol 2009;57:567-84. [PubMed]
- Finn AV, Nakazawa G, Joner M, et al. Vascular responses to drug eluting stents: importance of delayed healing. Arterioscler Thromb Vasc Biol 2007;27:1500-10. [PubMed]
- Kolandaivelu K, Swaminathan R, Gibson WJ, et al. Stent thrombogenicity early in high-risk interventional settings is driven by stent design and deployment and protected by polymer-drug coatings. Circulation 2011;123:1400-9. [PubMed]
- Briguori C, Sarais C, Pagnotta P, et al. In-stent restenosis in small coronary arteries: impact of strut thickness. J Am Coll Cardiol 2002;40:403-9. [PubMed]
- Jabara R, Geva S, Ribeiro HB, et al. A third generation ultra-thin strut cobalt chromium stent: histopathological evaluation in porcine coronary arteries. EuroIntervention 2009;5:619-26. [PubMed]
- Ribeiro EE, Campos CM, Ribeiro HB, et al. First-in-man randomised comparison of a novel sirolimus-eluting stent with abluminal biodegradable polymer and thin-strut cobalt-chromium alloy: INSPIRON-I trial. EuroIntervention 2014;9:1380-4. [PubMed]
- Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344-51. [PubMed]
- Pache J, Kastrati A, Mehilli J, et al. Intracoronary stenting and angiographic results: strut thickness effect on restenosis outcome (ISAR-STEREO-2) trial. J Am Coll Cardiol 2003;41:1283-8. [PubMed]
- Kastrati A, Mehilli J, Dirschinger J, et al. Intracoronary stenting and angiographic results: strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation 2001;103:2816-21. [PubMed]
- Basalus MW, Joner M, von Birgelen C, et al. Polymer coatings on drug-eluting stents: Samson's hair and Achilles' heel? EuroIntervention 2013;9:302-5. [PubMed]
- Mehilli J, Byrne RA, Wieczorek A, et al. Randomized trial of three rapamycin-eluting stents with different coating strategies for the reduction of coronary restenosis. Eur Heart J 2008;29:1975-82. [PubMed]
- Byrne RA, Kastrati A, Kufner S, et al. Randomized, non-inferiority trial of three limus agent-eluting stents with different polymer coatings: the Intracoronary Stenting and Angiographic Results: Test Efficacy of 3 Limus-Eluting Stents (ISAR-TEST-4) Trial. Eur Heart J 2009;30:2441-9. [PubMed]
- Windecker S, Serruys PW, Wandel S, et al. Biolimus-eluting stent with biodegradable polymer versus sirolimus-eluting stent with durable polymer for coronary revascularisation (LEADERS): a randomised non-inferiority trial. Lancet 2008;372:1163-73. [PubMed]
- Stefanini GG, Byrne RA, Serruys PW, et al. Biodegradable polymer drug-eluting stents reduce the risk of stent thrombosis at 4 years in patients undergoing percutaneous coronary intervention: a pooled analysis of individual patient data from the ISAR-TEST 3, ISAR-TEST 4, and LEADERS randomized trials. Eur Heart J 2012;33:1214-22. [PubMed]
- de Waha A, King LA, Stefanini GG, et al. Long-term outcomes of biodegradable versus durable polymer drug-eluting stents in patients with acute ST-segment elevation myocardial infarction: a pooled analysis of individual patient data from three randomised trials. EuroIntervention 2015;10:1425-31. [PubMed]
- Palmerini T, Biondi-Zoccai G, Della Riva D, et al. Clinical outcomes with bioabsorbable polymer- versus durable polymer-based drug-eluting and bare-metal stents: evidence from a comprehensive network meta-analysis. J Am Coll Cardiol 2014;63:299-307. [PubMed]
- Lemos PA, Moulin B, Perin MA, et al. Rationale and design for the PAINT randomized trial. Arq Bras Cardiol 2009;93:547-53, 590-7. [PubMed]
- Lemos PA, Moulin B, Perin MA, et al. Randomized evaluation of two drug-eluting stents with identical metallic platform and biodegradable polymer but different agents (paclitaxel or sirolimus) compared against bare stents: 1-year results of the PAINT trial. Catheter Cardiovasc Interv 2009;74:665-73. [PubMed]
- Lemos PA, Moulin B, Perin MA, et al. Late clinical outcomes after implantation of drug-eluting stents coated with biodegradable polymers: 3-year follow-up of the PAINT randomised trial. EuroIntervention 2012;8:117-9. [PubMed]
- Marchini JF, Gomes WF, Moulin B, et al. Very late outcomes of drug-eluting stents coated with biodegradable polymers: insights from the 5-year follow-up of the randomized PAINT trial. Cardiovasc Diagn Ther 2014;4:480-6. [PubMed]


